What is the value of compression factor Z for the gas? (A) 1 (B) >1 (C) <1 (D) Zero

By A Mystery Man Writer

Solved (Triple-Play Bonus) For a certain gas, the

Physical Chemistry The Compression Factor (Z) [w/1 example
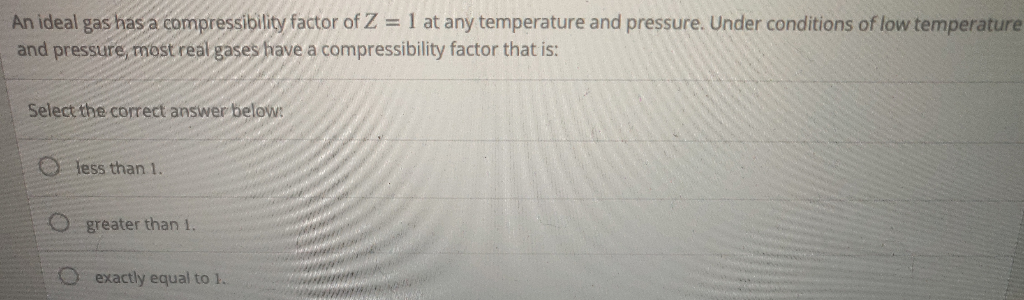
Solved An ideal gas has a compressibility factor of Z = 1 at

W 80 4 0 hi-res stock photography and images - Page 3 - Alamy

The compression factor (compressibility factor) for `1 mol` of a

gas laws - Graph of compressibility factor vs pressure when real
The value of compression factor at the critical state of a vander

Compressibility factor, Z of a gas is given as Z = pV / nRTi What

Compression Factor Z

Chapter 1. Properties of Gases - ppt download
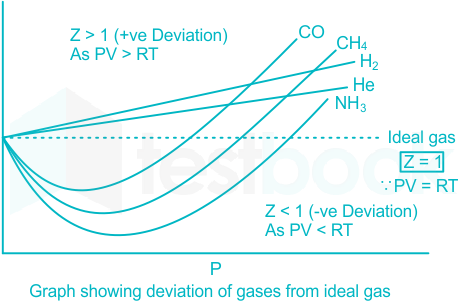
Solved] The compressibility factor for an ideal gas is

Problems in general physics by Mateus Pereira - Issuu

Gas compressibility factor Z: Ideal gas vs Real gas

Teachers book physics1 1 by Nick Ioannou - Issuu
- What is the compression ratio, and how is it calculated? - Quora
- Derive an expression for the compression factor of a gas tha
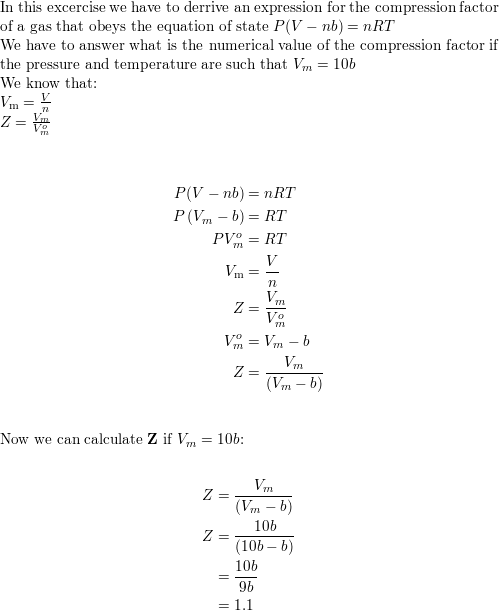
- Solved 6. Let's see if we can derive an alternative
- The Compression Factor, Z, and Real Gases - What you NEED to Know!

- SOLVED: For a gas at a given temperature, the compression factor is described by the empirical equation: z = 1 - 8.50 × 10^(-3)P/P° + 3.50 × 10^(-5)(P/P°)^2 where P° = 1






