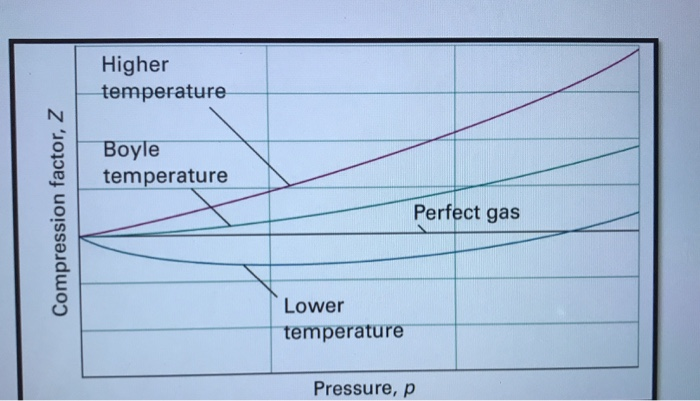
Consider the graph between compressibility factor Z and pressure P

By A Mystery Man Writer
Z1 means force of attraction dominating ie a is considerable b can be negligible at low temperature and low pressure Lower is the value of Z easier is the process of liquification
The compressibility factor is actually a factor that corrects the actual value of the gas versus the ideal gas. Let us learn and understand this concept.
Watch this video to understand the behaviour of real gases with the help of the compressibility factor. This is an important topic for JEE main.
What is the compressibility factor, and how does it vary with an increase in temperature and pressure? Watch this video to get the answer. This is an importa

Consider the graph between compressibility factor Z and pressure P

The electrophile involved in above reaction has lone pair of electrons
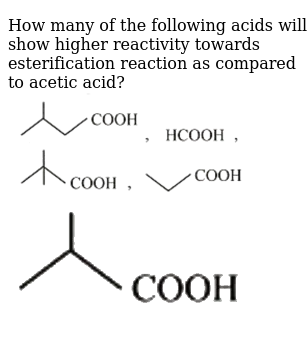
How many of the following acids will show higher reactivity towards es
Solved 6. (a) Discuss the significance of the Boyle

Chemistry Desk: Effect of Pressure

How many of the following acids will show higher reactivity towards es

Compressibility factor of benzene vapor along its saturation curve. P i

1. Consider a fluid that obeys the van der Waals

Consider the graph between compressibility factor Z and pressure P
- Compressibility factor, Z of a gas is given as Z= frac { pV }{ nRT } (i) What is the value of Z an ideal gas?(ii) For real gas what will be

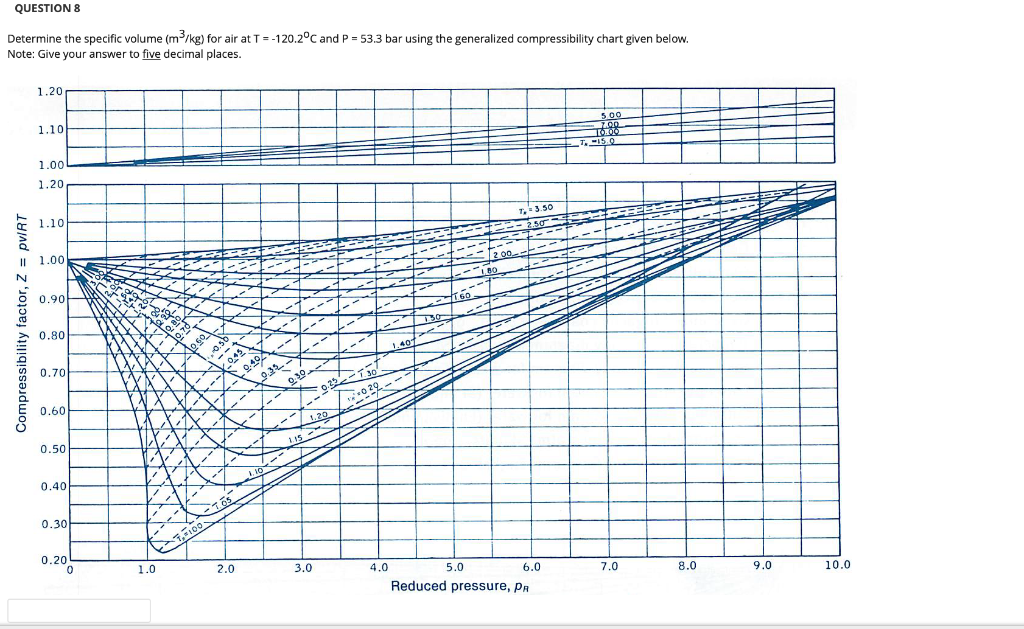
- Solved QUESTION 3 Determine the compressibility
- Determine Compressibility Factor with Present of CO2 and H2S
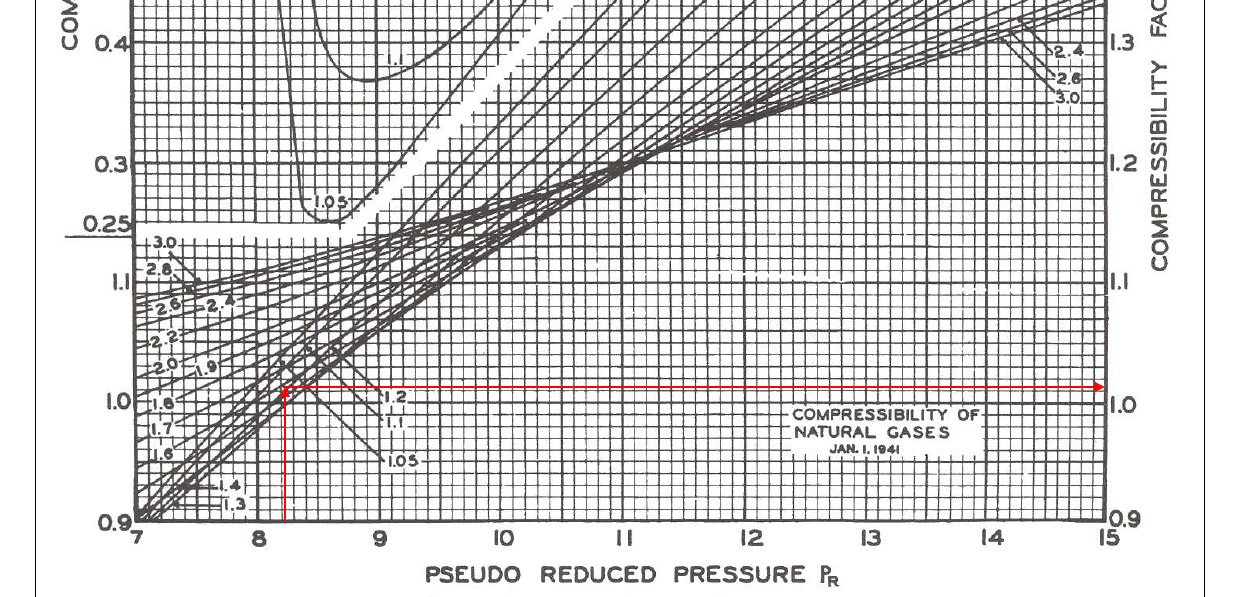
- For a given gas, a graph is shown between compressibility factor

- Compressibility factor of water
- Glamorise 1000 Magic Lift Bra

- Off-White set 2 slip in cotone stretch donna - Glamood Outlet

- Summer Sweatpants For Women: Solid Color Capri Terry Cropped Trouser With Elastic Waist Pockets And Wide Leg From Onlywear, $21.07

- Absolute Territory Knee Socks/Thigh High - Up LoRA - PromptHero

- Maidenform Women's Maidenform Custom Lift Strapless Bra,White,36A at Women's Clothing store: Bras
