thermodynamics - Variation of compressiblity factor with

By A Mystery Man Writer
While I certainly understand the order of temperatures, I can't find a reason for the curves to intersect at one common point. Why do the curves intersect at one point? or do they really intersect

The compressibility factor Z versus the temperature T [K], for 4 He

Gas compressibility factor Z: Ideal gas vs Real gas

Comparison of various state equations for carbon dioxide at 9 MPa

thermodynamics - Variation of compressiblity factor with temperature - Chemistry Stack Exchange

Deviation of Real Gases from Ideal Gas Behaviour - GeeksforGeeks

Gas compressibility factor Z: Ideal gas vs Real gas

Acentric Factor - an overview
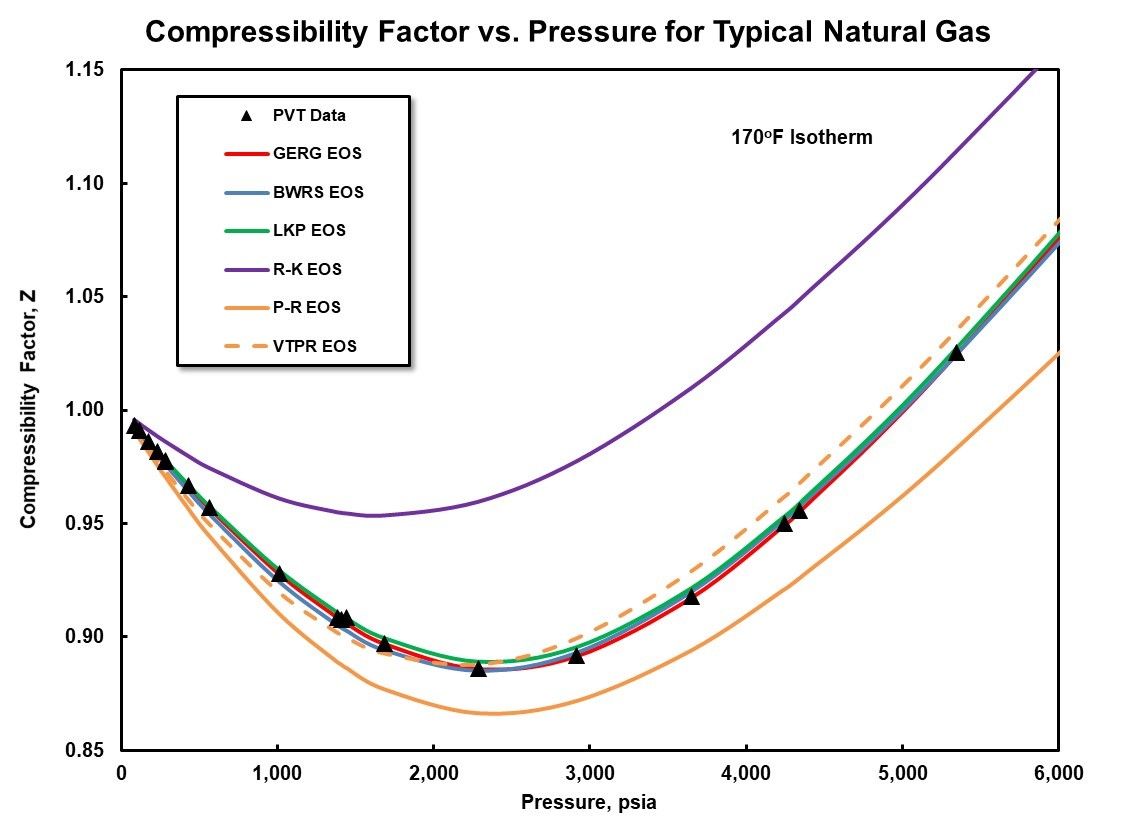
Compressor performance and thermodynamics

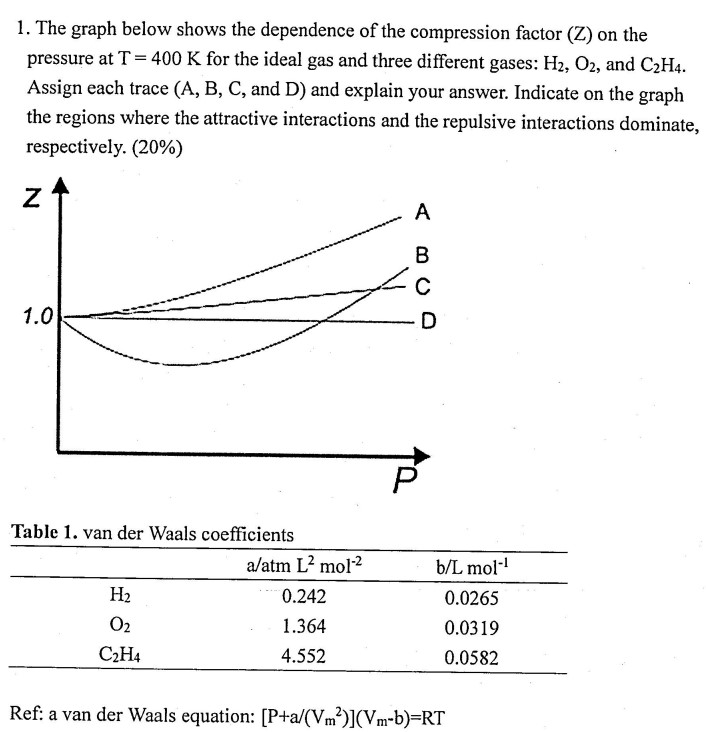
The given graph represents the variations of compressibility factor `Z=PV//nRT` vs `
- Buy Women Sexy High Waisted French Knickers Lace Panties Brief Stretchy Sheer Floral Underpants Underwear Online at desertcartZimbabwe

- Girl Lolita Heart Japanese Bra and Panty Set Plush Cat Paw Underwear Lovely Small Chest Gather Bra Set no Steel Ring Brassiere

- 40g 80ml PP Measure Spoon 40 Gram Plastic Measuring Scoop for Milk

- Period Swimwear Mid Waisted Bottoms Fuschia Pink – Savvi Wear

- eChai Ventures on X: .@eChaiVentures, @SGAhmedabad, @ihubgujarat


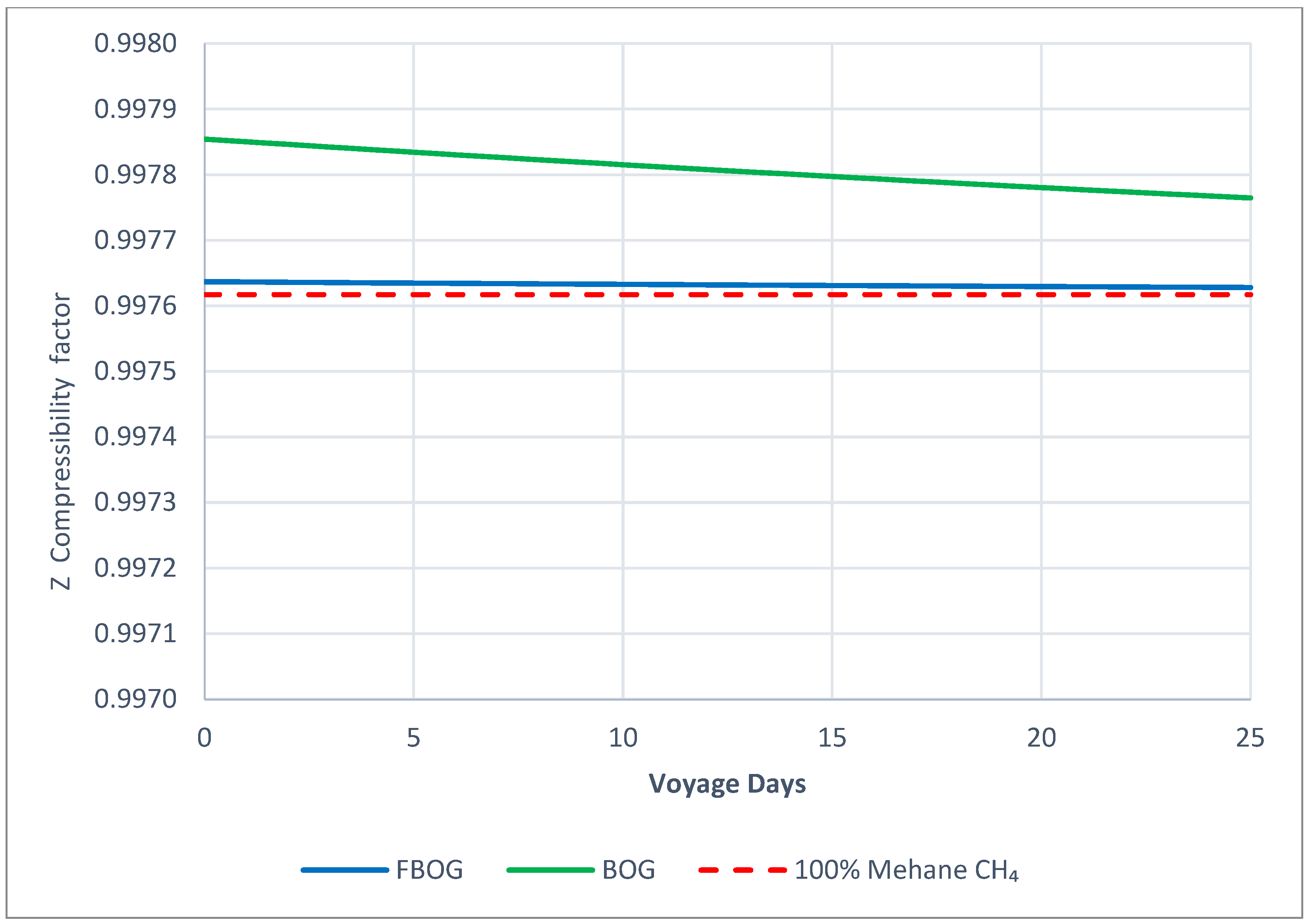
:max_bytes(150000):strip_icc()/Term-Definitions_zscore.aspsketch-3c68631c0479475f9bee1d6ecb0f4e23.jpg)
