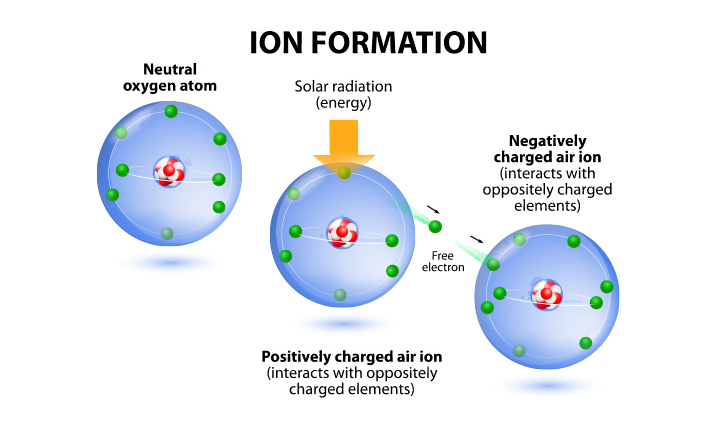
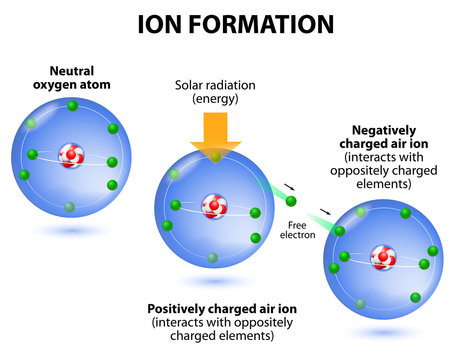
How to memorize positive ions and negative ions fast - Quora
By A Mystery Man Writer
Answer: If you have a periodic table, just remember that the vertical columns (groups) are arranged by their valence electrons. Group 1 has one valence electron, so it will likely lose one electron to stabilize its valence, giving Group 1 a 1+ charge. Group 16 has six valence electrons, so it wil

boAt Nirvana 525 ANC Bluetooth Neckband with Surround Sound by Dolby Audio, Adaptive EQ, Active Noise Cancellation, ENx Technology, 30 HRS Playback and ASAP Charge(Space Black) : : Electronics
What are the three methods of charging a body? - Quora
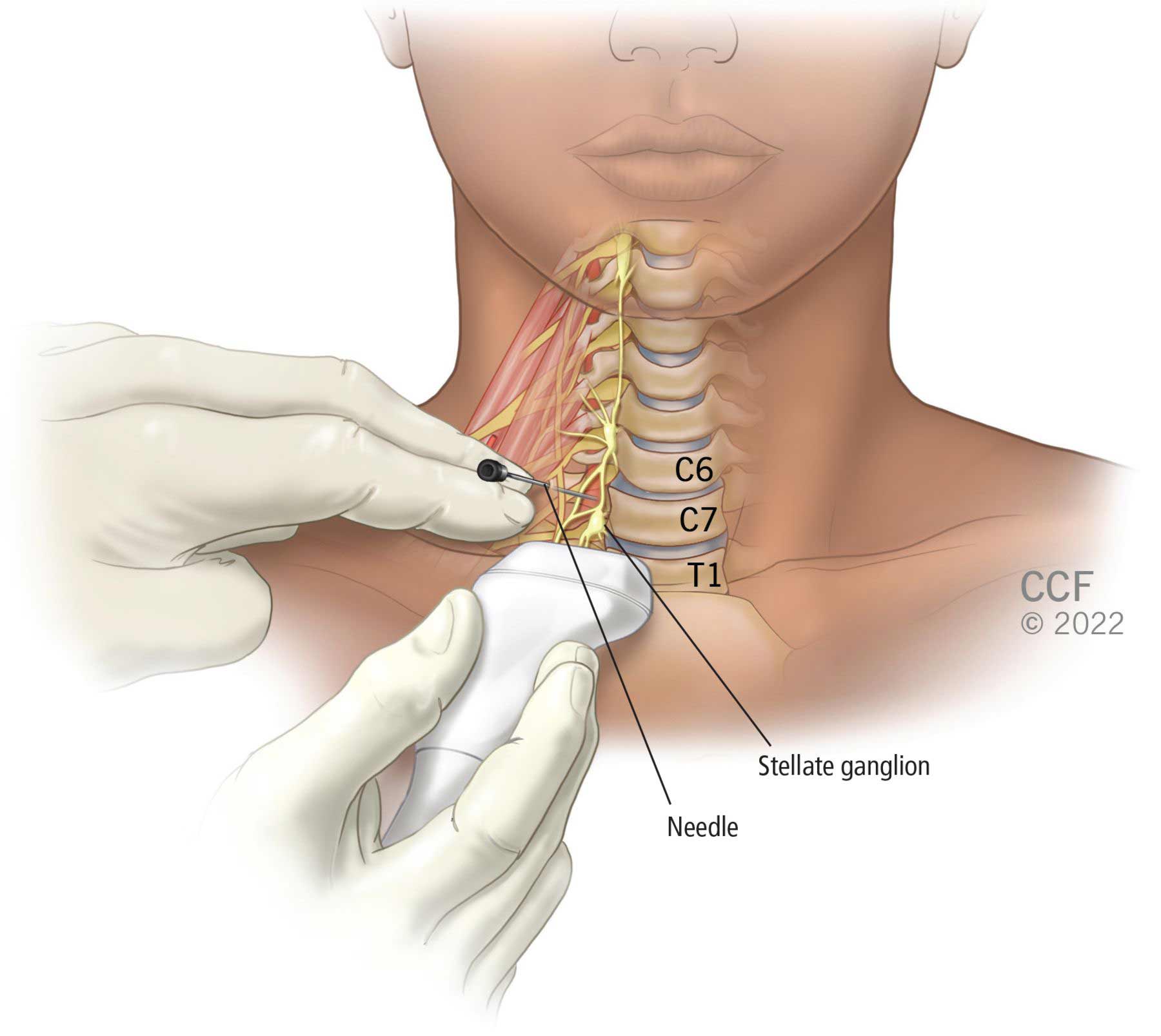
Stellate Ganglion Block: What It Is, Uses, Side Effects & Risks

CH104: Chapter 3 - Ions and Ionic Compounds - Chemistry
Is there any trick to learn the cation and anion tables? - Quora
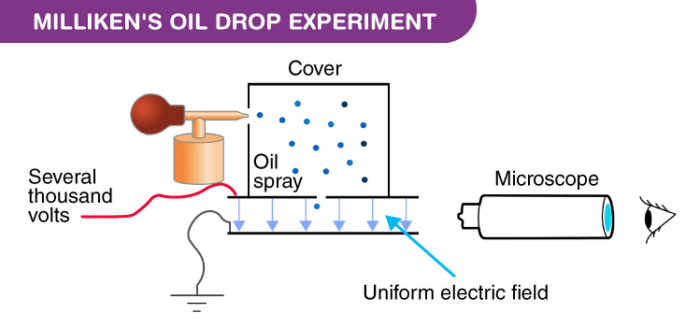
Millikens Oil Drop Experiment - Apparatus, Procedure Calculation & Conclusion

50 of the biggest sustainability questions answered - FutureLearn

KONE Corporation - Improving the Flow of Urban Life. - KONE Corporation

Key Question: Why Do Atoms Combine In Certain Ratios? - ppt download

KONE Corporation - Improving the Flow of Urban Life. - KONE Corporation
What is the best way of knowing if an atom will gain, or lose, an electron? - Quora
La Trobe University, Melbourne Victoria Australia