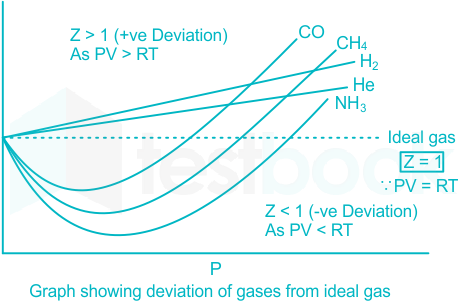
The given graph represent the variations of compressibility factor (z) = pV/nRT versus p, - Sarthaks eConnect
By A Mystery Man Writer
The given graph represent the variations of compressibility factor (z) = pV/nRT versus p, for three , the slope is positive for all real gases.
Solved] The compressibility factor for an ideal gas is
Pressure versus volume graph for a real gas and an ideal gas are

The given graph represents the variations of compressibility

DV (a) nb (c) – (n'a/v2) (d) - nb The given graph represent the

Thermodynamics 3-92
Deduce the relation pv = nRT where R is a constant called

Telugu] The variation of compressibility factor (Z) with pressure (p

Compressibility factor (Z=(PV)/(nRT)) is plotted against pressure

The graph of compressibility factor Z vs P for one mole of a real

The given graph represents the variation of Z(compressibility

The given graph represents the variation of Z(compressibility factor =- PV nRT ) versus P, three real gases A, B and C. Identify the only incorrect statement. Ideal gas P (atm) (A)

The given graph represents the variation of compressibility factor

The compressibility factor `(Z=PV//nRT)` for `N_(2)` at `223 K
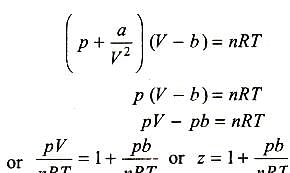
The given graph represents the variation of Z (compressibility
- Compressibility factor (gases) - Knowino

- At low pressure, the van der waal's equation is written as (P+ a/V^2)V=RT . Then compressibility factor is then equal to
- PPT - GASES PowerPoint Presentation, free download - ID:2088317

- 3.2 Real gas and compressibility factor – Introduction to

- Solved] Why is the compressibility factor less than 1 at most
- Womens Crop Top And Stacked Pants Club Outfit Set Baddie Funeral Outfit Women For Instagram Clothes 71284 From Dou02, $23

- ArtStation - SpyxFamily Yor Forger Bust

- Tighten Multiple Fasteners in One Quick and Easy Step - Metis Automation

- YWDJ Bras for Women Push Up for Large Bust Front Closure Zip Snap Show Up Front Close Sagging Breasts Lightly Front Buckle Upper Big Chest Show Small

- NYDJ - Cropped Hoodie – Beyond Marketplace
