At a given temperature T gases Ne Ar Xe and Kr are found to

By A Mystery Man Writer
For a certain gas which deviates a little from ideal behaviour , a plot between P/d vs P was found to be non linear . The intercept on y axis will be . (

Oswaal IIT JEE Main Solved Papers Chemistry Chapterwise and Topicwise 2019 and 2020 All shifts 32 Papers

Characteristic times, τ c , for noble gases versus temperature, T, of

SOLVED: A 1.07 g sample of a Noble gas occupies a volume of 363 mL at 35*C and 0.892 atm. Identify the Noble gas in this sample. He Ne Ar Kr Xe

At a given temperature T, gases Ne,Ar,Xe and Kr are found to deviate from..
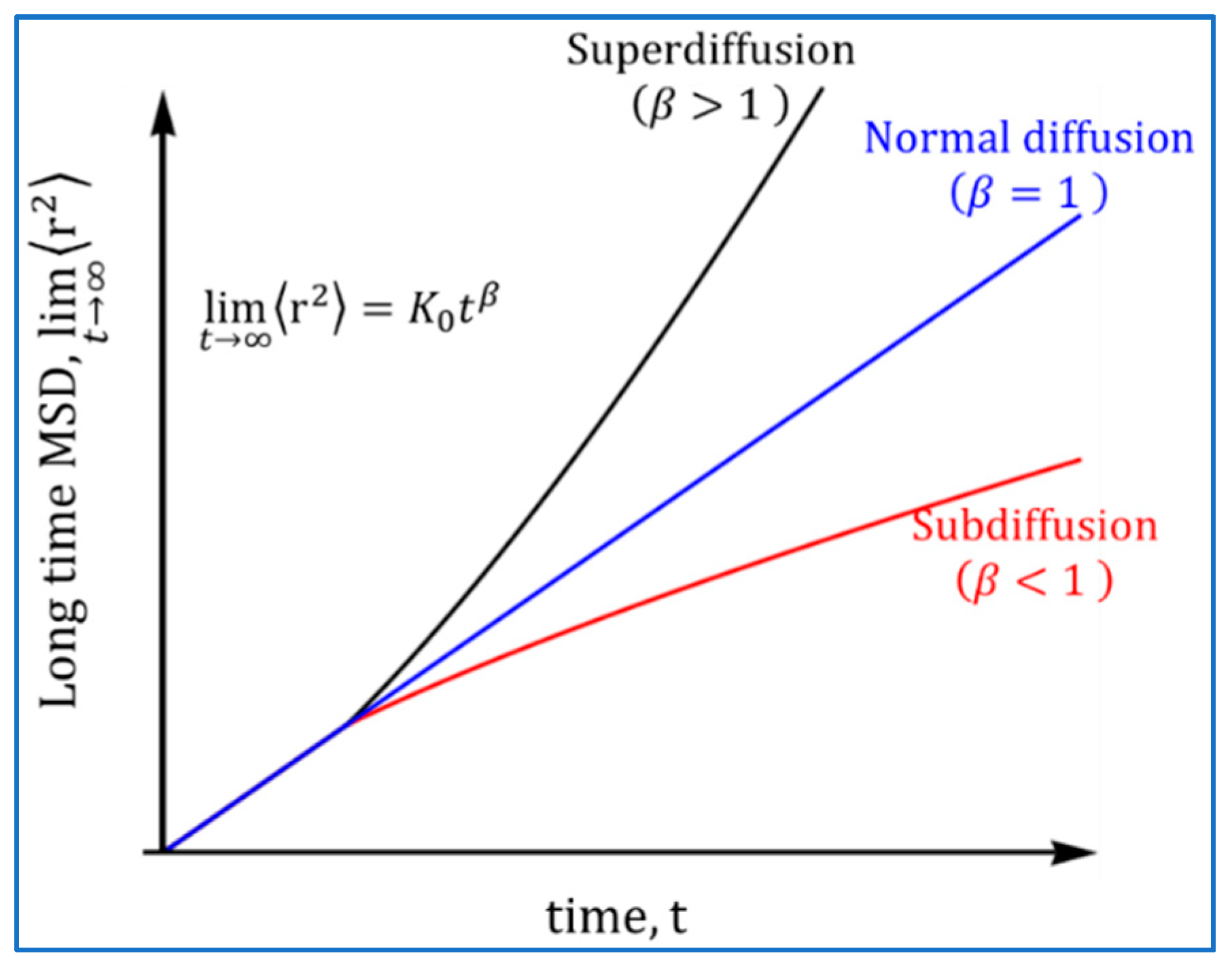
Processes, Free Full-Text

At a given temperature T, gases Ne, Ar, Xe and Kr are found to deviate from ideal gas behaviour. Their equation of state is given as p = dfrac {RT}{V - b}

⏩SOLVED:At a given temperature T, gases Ne, Ar, Xe and Kr are found…
Potential energy curves of Ne-Ar, Ne-Kr, Ne-Xe, Ne-Rn, Ar-Kr, Ar-Xe

The rate of diffusion of methane at a given temperature is twice that

A gas obeys the equation of state P(V-b) =RT (The parameter b is a con
- Solved 6. Let's see if we can derive an alternative
- COMPRESSION AND EXPANSION OF GASES – Chemical Engineering Projects

- Compression Factor Exam Problem using Molar Volumes - Fully Explained!

- Compared with the graph of the parent function, which equation shows only a vertical compression by a

- If `Z` is a compressibility factor, van der Waals' equation at low





