Medtronic Gains FDA Approval for Newest Insulin Pump - MedCity News

By A Mystery Man Writer
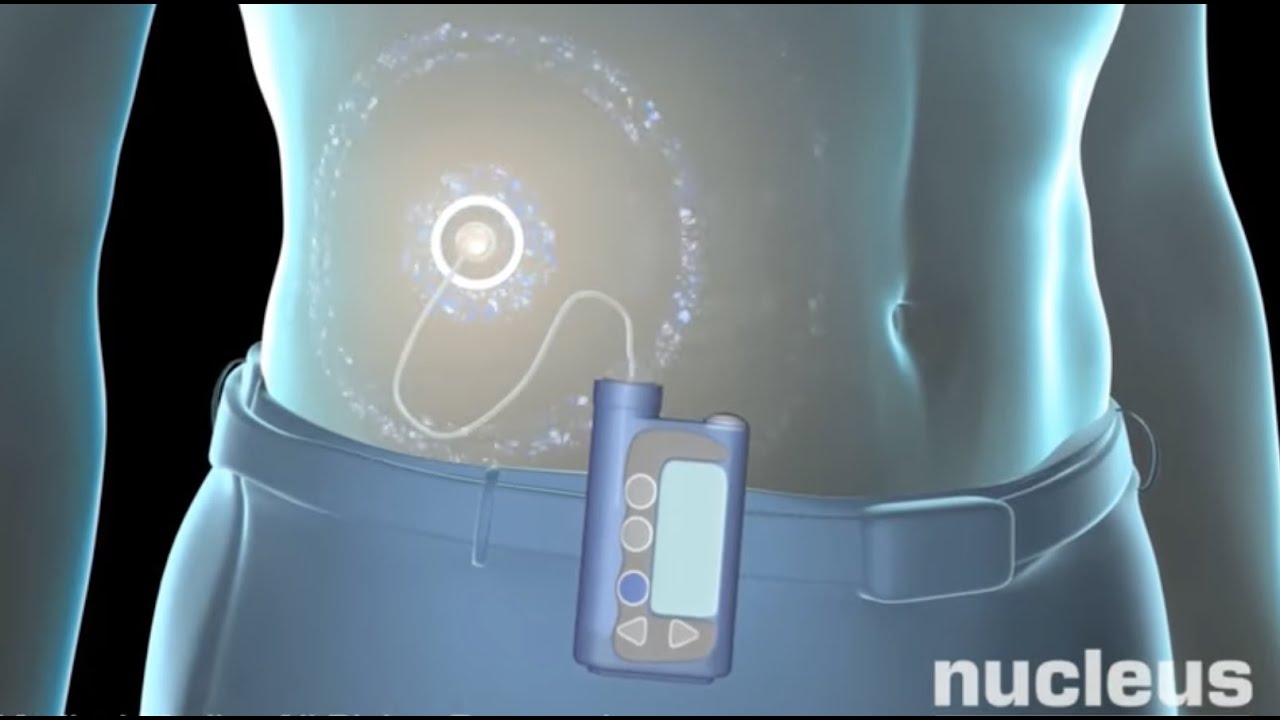
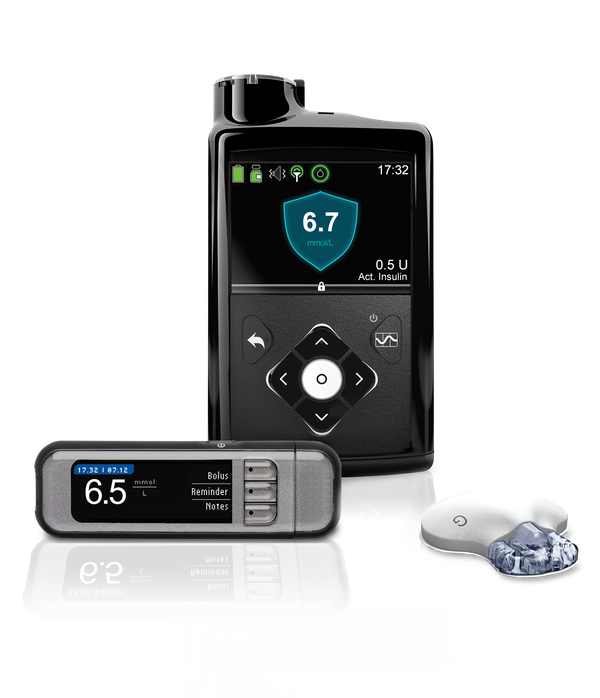
Medtronic recently received FDA approval for its MiniMed 780G system, which is an insulin pump that automatically adjusts and corrects type 1 diabetes patients’ glucose levels every five minutes. The news comes two years after Medtronic submitted the system for FDA approval and three years after the company began selling the product in Europe.
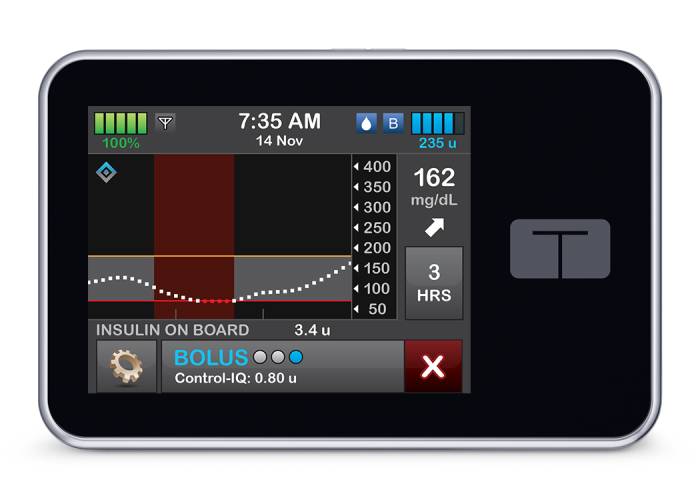
Tandem Diabetes Care - Recent News & Activity
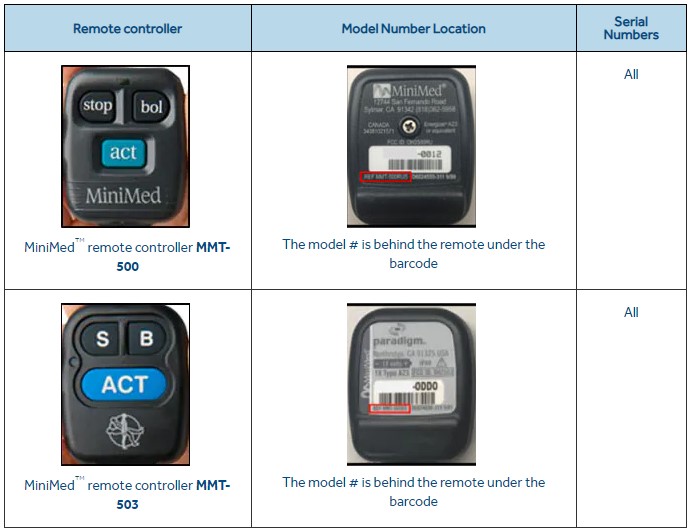
Medtronic urgently recalls insulin pump controllers over hacking

Tandem Diabetes Care - Recent News & Activity

Type 1 diabetes Archives - MedCity News

Medtronic MiniMed 780G Now Available In The US, Covered By, 59% OFF

Medtronic makes further moves into Type 2 diabetes with FDA approval of new CGM sensor - MedCity News

NN-1471 - Drug Targets, Indications, Patents - Synapse

Medtronic scores European green light to combine its latest

FDA approves Medtronic's MiniMed 780G insulin pump

50 Best Medical Device and MedTech News Sites, Blogs – Pannam

Type 1 diabetes Archives - MedCity News

insulin pump Archives - MedCity News
Medtronic MiniMed Insulin Pump Lawsuit & Recall

Radical data interoperability picking up speed [survey]