Draft Guidance Document: Applications for Medical Device Investigational Testing Authorizations

By A Mystery Man Writer
This draft guidance document reflects Health Canada’s current thinking on Investigational Testing Authorizations (ITA) for medical devices and may be subject to changes as policy develops. The document clarifies application requirements and processes, including pre-ITA meetings, format for an ITA application and filing requests for revisions to an ITA.
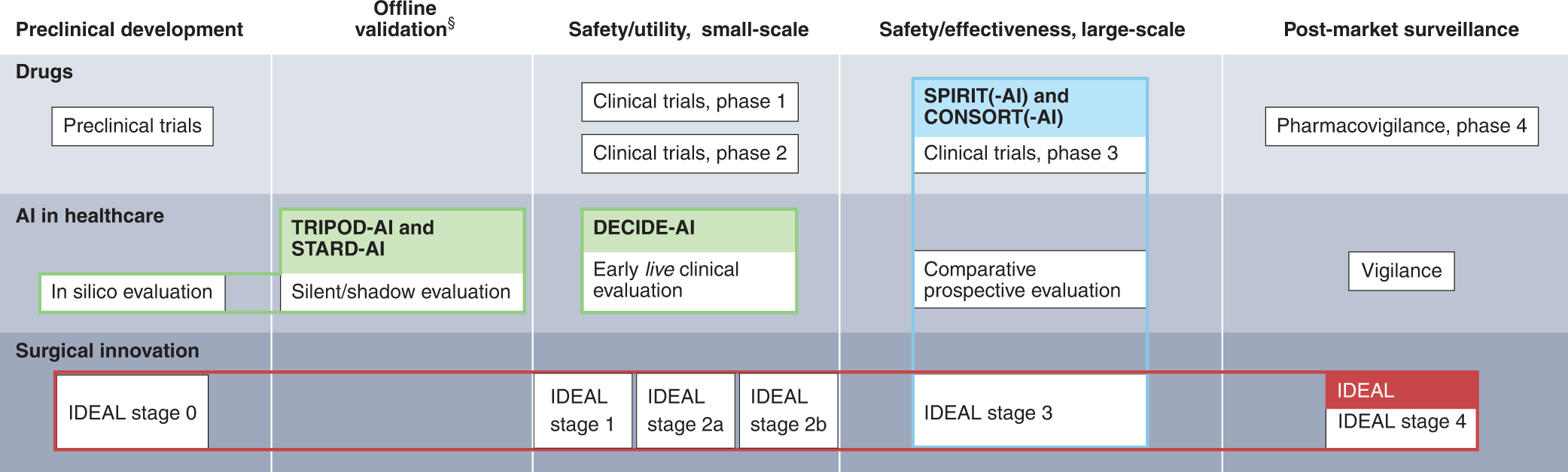
Reporting guideline for the early-stage clinical evaluation of decision support systems driven by artificial intelligence: DECIDE-AI

Multi-Society Consensus Conference and Guideline on the Treatment of Gastroesophageal Reflux Disease (GERD) - A SAGES Publication

FDA's Regulatory Framework for 3D Printing of Medical Devices at the Point of Care Needs More Clarity

Demystifying The Investigational Device Exemption Process - Healthcare - United States

Canada's Health Canada - Global Regulatory Partners, Inc.
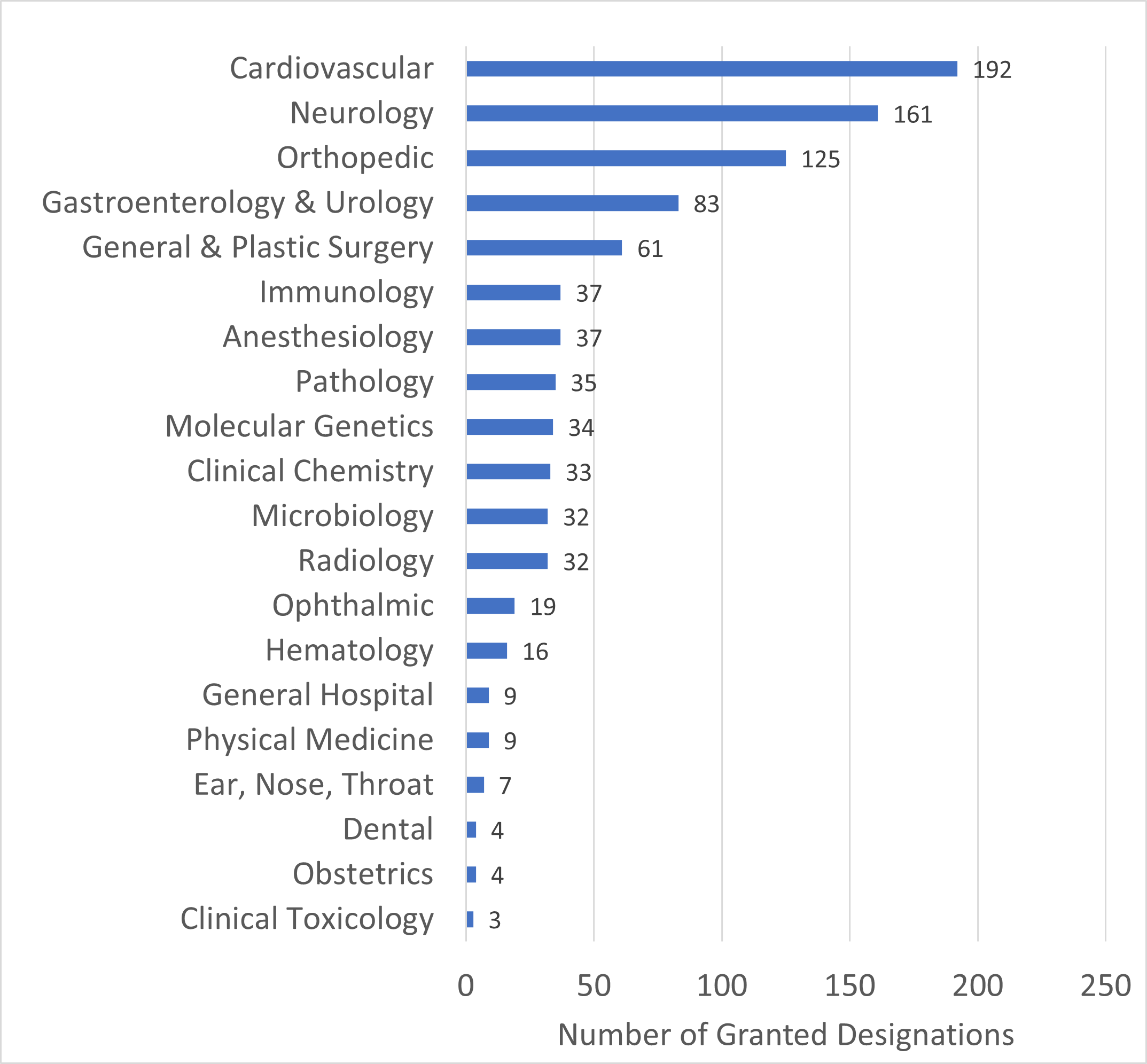
Breakthrough Devices Program
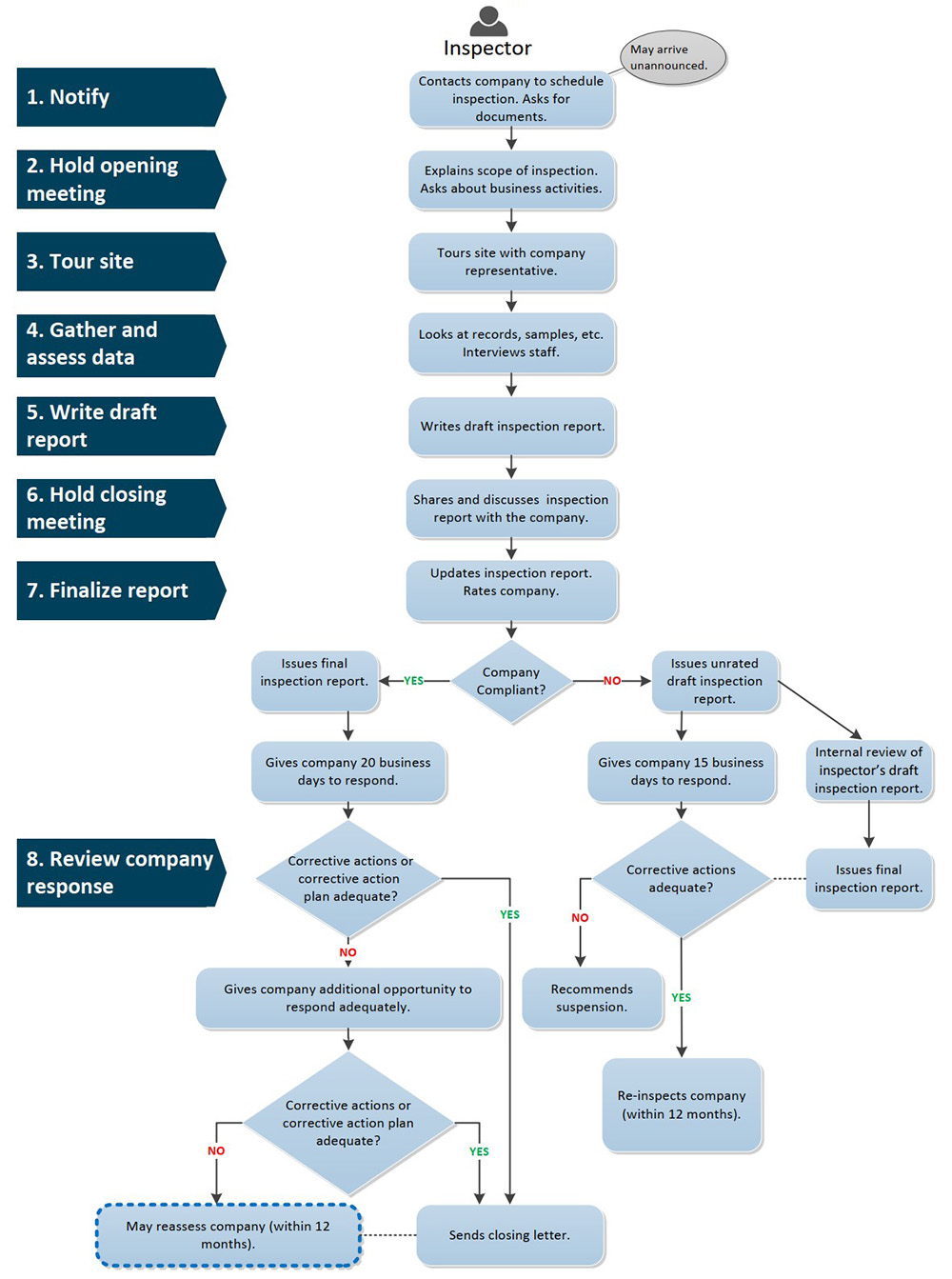
How Health Canada inspects medical device establishments: About inspections

Class II - IV Medical Device Investigational Testing in Canada - Vantage BioTrials

Emergency Use Authorization
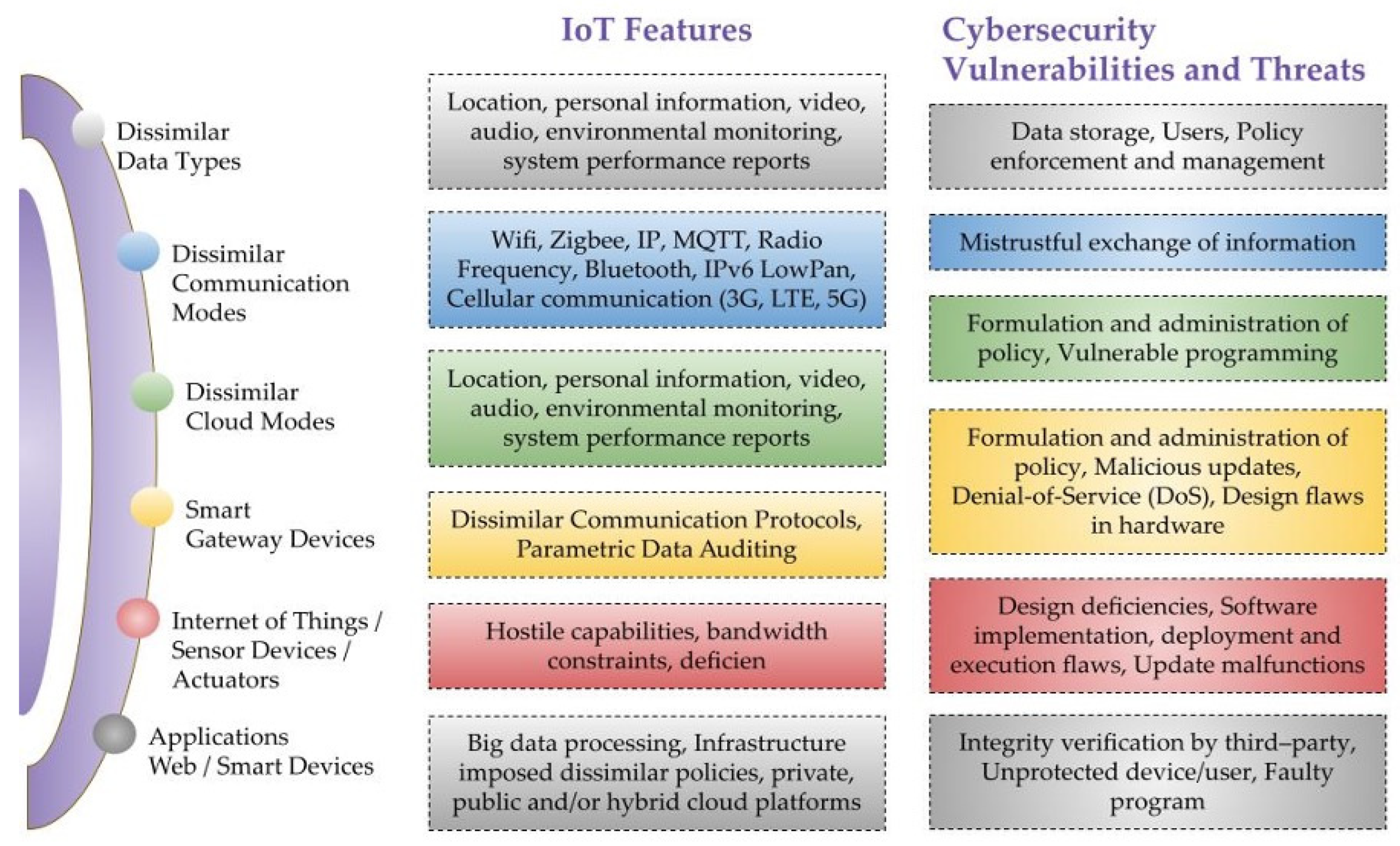
Sensors, Free Full-Text

Guideline for the production and quality control of monoclonal antibodies and related products intended for medicinal

Guidance on how to complete the application for a new medical device licence: Overview

Search for FDA Guidance Documents
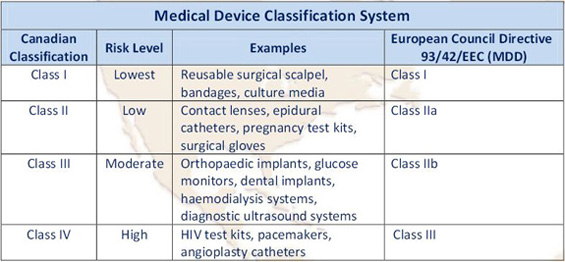
Class II - IV Medical Device Investigational Testing in Canada - Vantage BioTrials









