The compression factor (Z) Co, 7°C and 100 atm is 0.21. Calculate

By A Mystery Man Writer
Click here:point_up_2:to get an answer to your question :writing_hand:the compression factor z for co at 7c and 100atm is 021 calculate the volume
Click here👆to get an answer to your question ✍️ The compression factor -Z- Co- 7-C and 100 atm is 0-21- Calculate the volume of a 4 mole sample of co- same temperature and pressure -use R - 0-08 L- atm-K-mol -1- 0-192 -2- 0-05 L -3- 0-38 L -4- 0-44 L closed container can be

The compression factor (Z) for CO, at 7C and 100 atm is 0.21

Compressibility Factor Z

The compression factor (Z) Co, 7°C and 100 atm is 0.21. Calculate

Chemical Thermodynamics

The compression factor (Z) for CO2 at 7∘C and 100 atm is 0.21

Processes, Free Full-Text
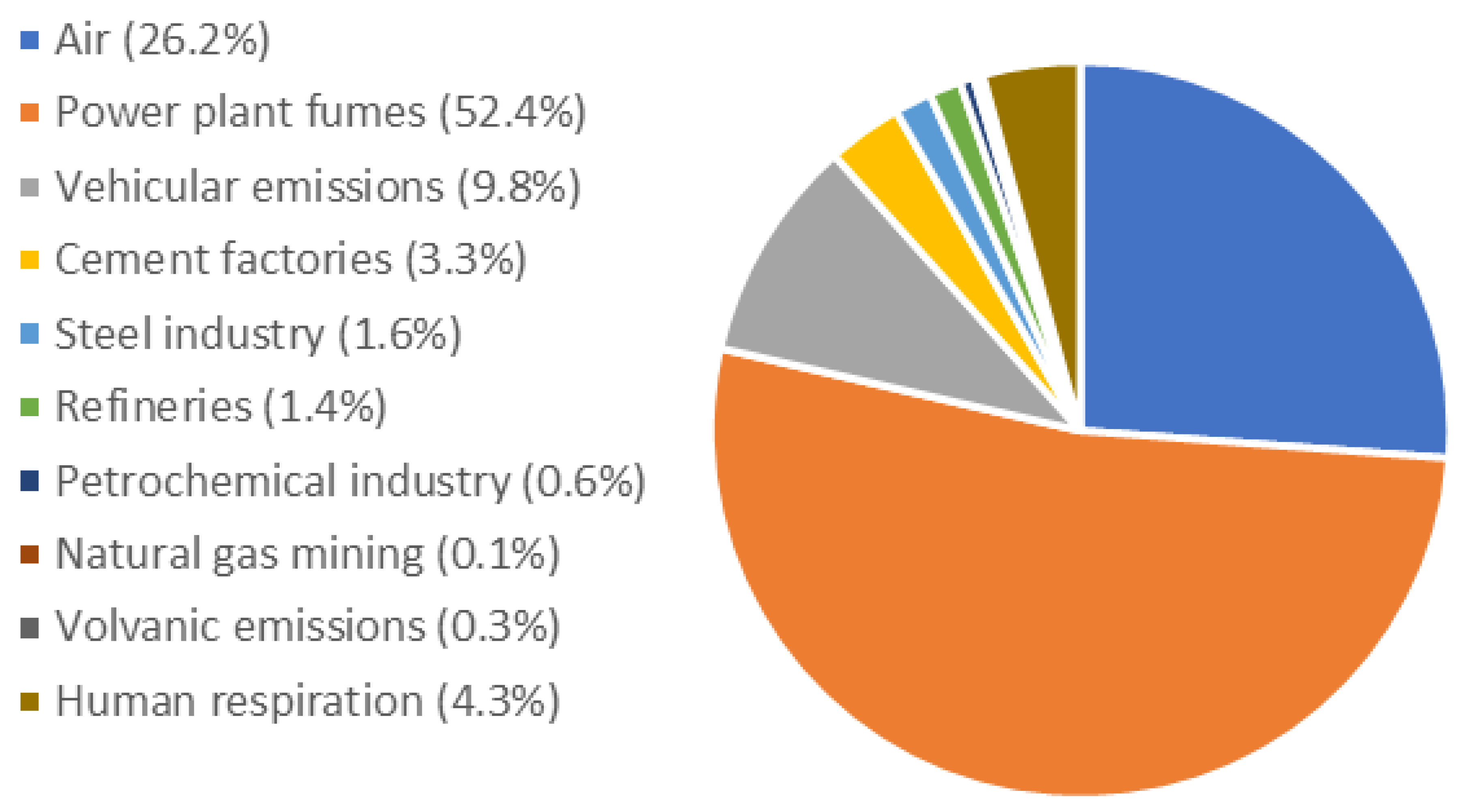
Atmosphere, Free Full-Text

Explosions of Syngas/CO2 Mixtures in Oxygen-Enriched Air

CH 6. State of Matter (Chem +1) PDF, PDF, Gases

Solved 1) The compression factor, Z, can be written as: Z
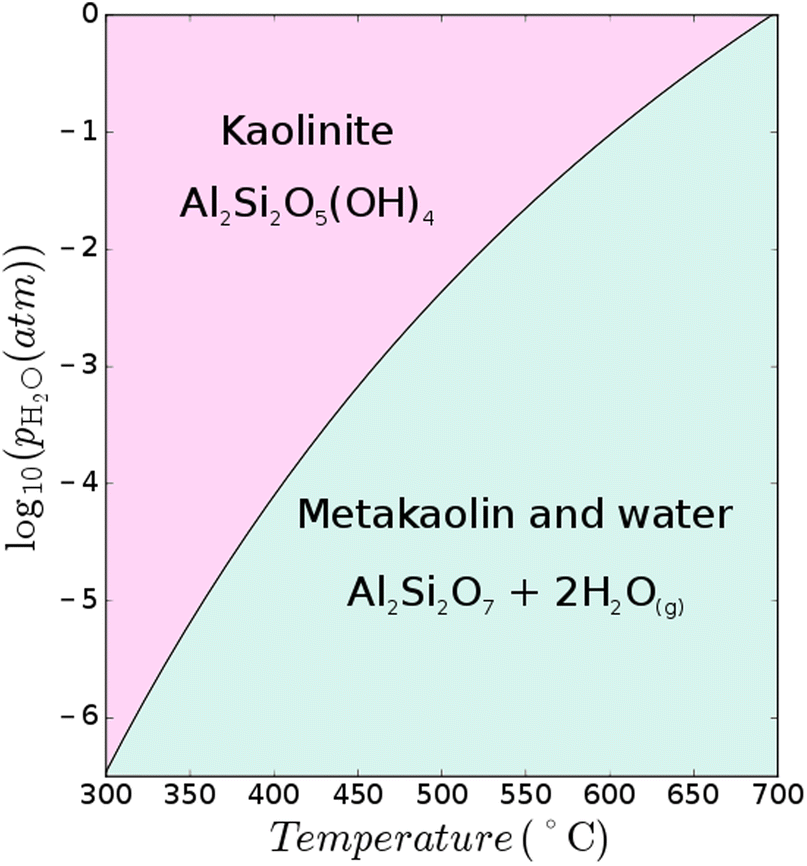
Thermodynamics of calcined clays used in cementitious binders


Physical Chemistry The Compression Factor (Z) [w/1 example

Acentric Factor - an overview
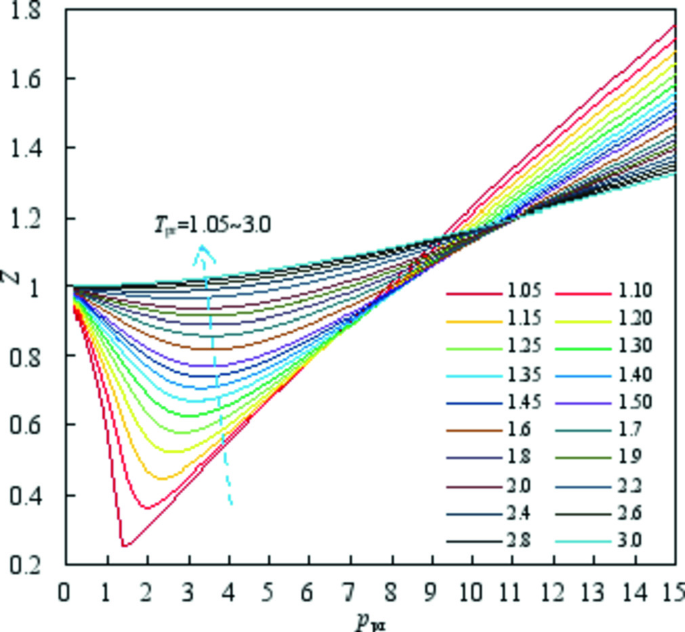
- Real gas z-factor, as attributed to Standing and Katz, 9 plotted as a

- gas laws - How to find the temperature relationship between the isotherms in a compressibility factor (Z) vs pressure graph? - Chemistry Stack Exchange

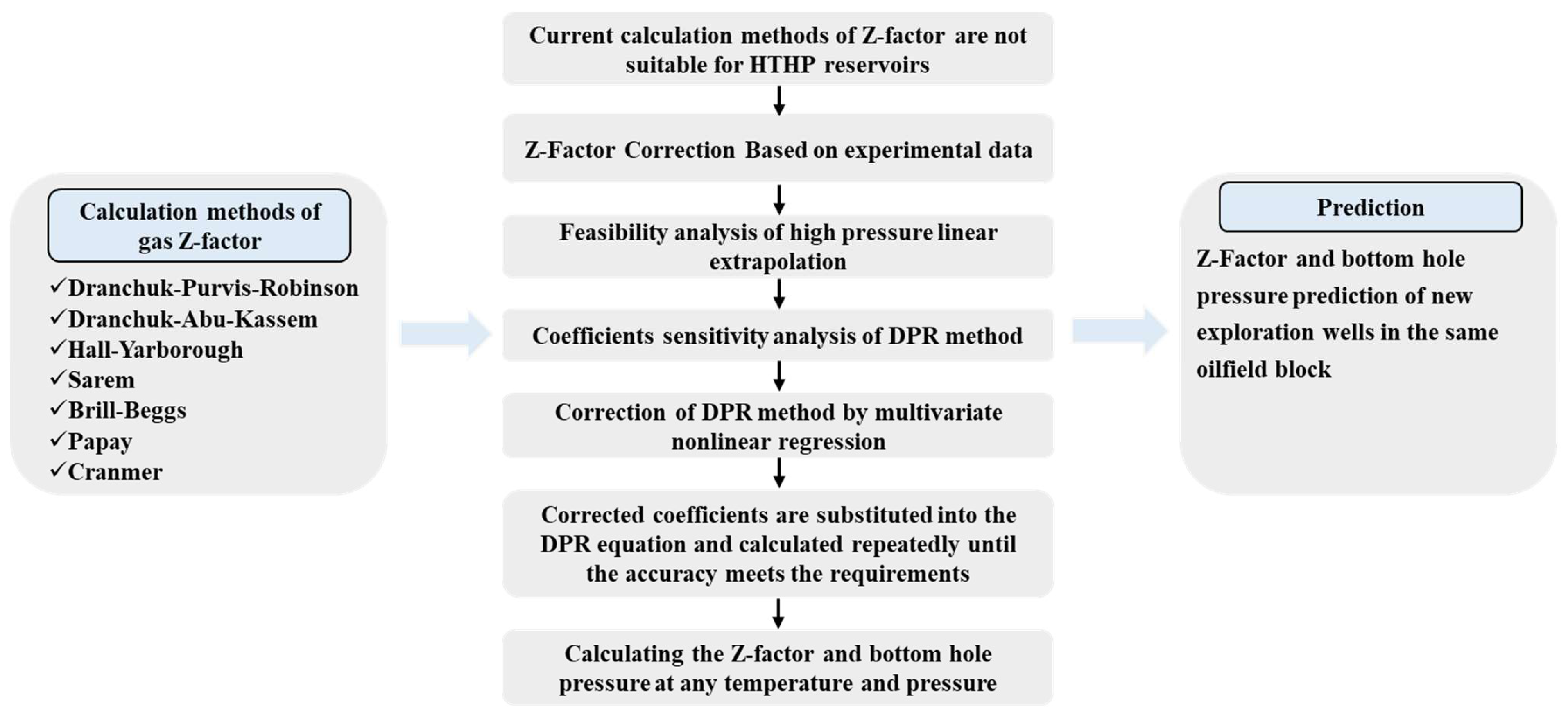
- Energies, Free Full-Text
- Calculating a Z-factor to assess the quality of a screening assay
.png)
- A New Method for Estimating Compressibility Factors of Natural
- NWT Delimira Burgundy Unlined Comfort Full Coverage Wire Free Bra

- TrainingGirl Corsé reductor de cintura para mujer, para pérdida de peso, cinturón de entrenamiento, banda para sudar, faja deportiva, traje de sauna – Yaxa Colombia

- Fall Wedding Guest Dresses for Women Over 40 — THE DAILEIGH

- Ashtanga Yoga Workshop - Ashtanga Yoga Satya

- Frozen Potty Training : Page 4 : Target
