Solved A diprotic base (B) has pKb values of 3.08 (pKb1) and

By A Mystery Man Writer

Docdownloader Com 17-Petrucci10e-Csm PDF, PDF, Titration

OneClass: The pKb values for the dibasic base B are pKb1 = 2.10 and pKb2 =7.85. Calculate the pH at e

Solved A diprotic base (B) has pKb values of 4.95(pKb1) and

SOLVED: A diprotic base B has pKa values of 3.54, 7.08, and 10.46. Which of the equalities is true when the pH is 6.92? [BH+] = [B] [BH] = [B] [BH+] = [

Solved A diprotic base (B) has pKb values of 4.95(pKb1) and

OneClass: A weak base (B) has a pKb value of 5.77. a) At what pH is [BH ] = [B]? b) What is the predo

All 1B CSM PDF, PDF, Reaction Rate

Chemistry of the Non-Metals: Syntheses - Structures - Bonding - Applications 9783110578058, 9783110578065, 9783110578317

OneClass: The pKb values for the dibasic base B are pKb1 = 2.10 and pKb2 =7.85. Calculate the pH at e

OneClass: The pKb values for the dibasic base B are pKb1 = 2.10 and pKb2 =7.85. Calculate the pH at e

PH Adjuster and Buffering, PDF, Buffer Solution
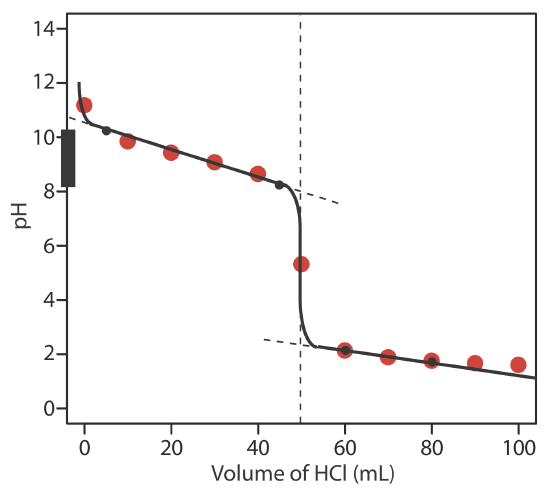
6.6: pH Calculations for Acid–Base Titrations - Chemistry LibreTexts
- TALLY Bra Set Ada Kawat Bahan Stretch Pori Busa Tipis Cup B Polos BH 304 / 2210

- Bh beugel licht push up effect zwart naadloze cup Wacoal - Bodyfashion Born

- What is the difference between BH and MH curves in magnetism? - Quora
- Bh Logo Images – Browse 1,881 Stock Photos, Vectors, and Video

- B-H curves of ferrite core: (a) dc magnetic property; (b) ac

- Madewell Strawberry Embroidered (Re) sourced Cotton Shrunken Shirttail Tee Lighthouse XS (Women's 00-0) at Women's Clothing store

- Capezio Women's Ultra Soft Stirrup Body Tight : Target
- Under Armour ua w hovr phantom nc Zapatilla de Mujer Zapatillas hombre moda, Zapatillas mujer, Calzado masculino

- Brandy melville black mabel - Gem

- Dark Orange Halloween Fairy Dance Costume Rave Bra and Skirt – L'Amour Le Allure
