The value of compression factor at the critical state of a vander
By A Mystery Man Writer
The value of compression factor at the critical state of a vander waals gas is
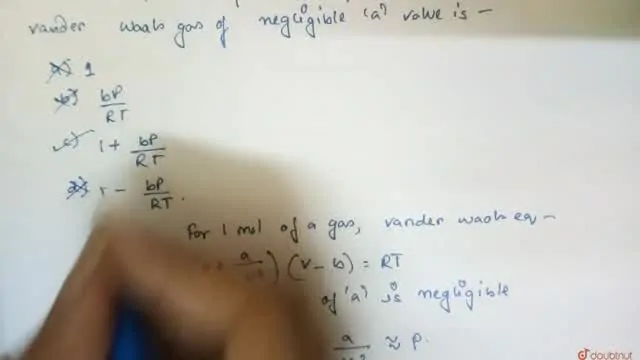
Bengali] The compresibility factor (Z) of one mole of a van der waals

SOLVED: 1) Estimate/ Calculate the critical constants (pc, Vc, and Tc) for a gas molecule whose van der Waals parameters are a = 1.32 atm dm^6 mol^-2 and b = 0.0436 dm^3

Solved Problem 1. Let's consider the van der Waals equation

The value of compression factor at the critical state of a vander waals gas is

The value of compressibility factor at the critical state the gas matc
Gaseous State.pdf - Chemistry - Notes - Teachmint

Physical Chemistry The Compression Factor (Z) [w/1 example]

The value of compressibility factor at the critical state the gas matc

Shear strength: Friction. a Critical state friction angle and particle
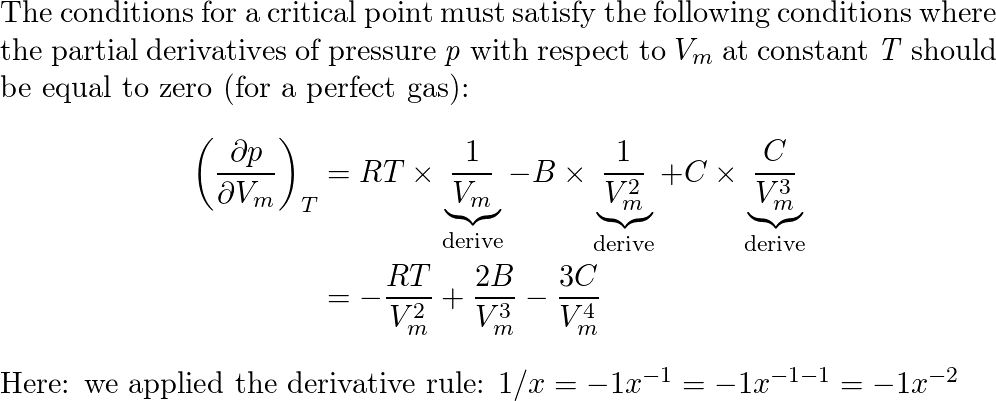
A scientist proposed the following equation of state $p= ra

Liquid-vapour saturation curve and Γ < 0 region (shaded region) for a

What is the value of z (compressibility factor) for a vander waal gas at critical
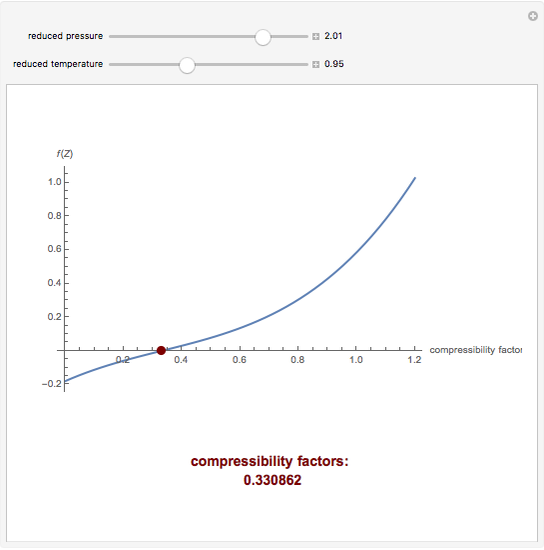
- Cubic Equation of State for the Compressibility Factor - Wolfram Demonstrations Project
- Compressor and jet vacuum system:, by Maryambotshekan

- Equation of state (excess compressibility factor, Z À1 ¼ PV/(NkT) À1

- Figure 3 from A Simple Equation Of State For Calculating The

- Non Ideal Gas Behavior-chemistry - Non Ideal Gas Behavior

- Fulani braids with quickweave😍😍✨🙌🏽 #fulanibraids #braids
- Nordstrom Sale: Grab Jennifer Aniston's Hanky Panky Panties

- 2pack Solid Underwire Bra

- Breathable Cool Lift Up Air Bra, Women's Seamless Air Permeable Cooling Comfort Bra, Breathable Comfort Air Bra : : Clothing, Shoes

- Womens Yoga Flare Pants Super Stretchy High Waist Leggings For Gym

