Tuesday, Oct 08 2024
Ideal–Universal Gas Law

By A Mystery Man Writer
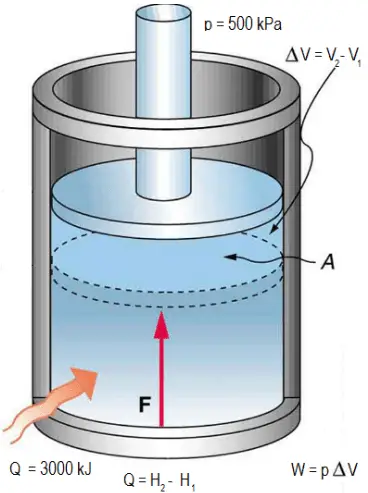
Definition: The Universal or Ideal Gas Law describes the relationship between all four properties (pressure, volume, number of moles, and temperature) as well as a gas constant called “R.” NOTE: The Ideal Gas Constant “R” has constant a value of 0.0821 L.atm/mol.K Relation: The relation between pressure (P) volume (V), number of moles (n) and…

Equation of State

Ideal Gas Equation - Infinity Learn by Sri Chaitanya

The Mole Concept: Mass Relationships
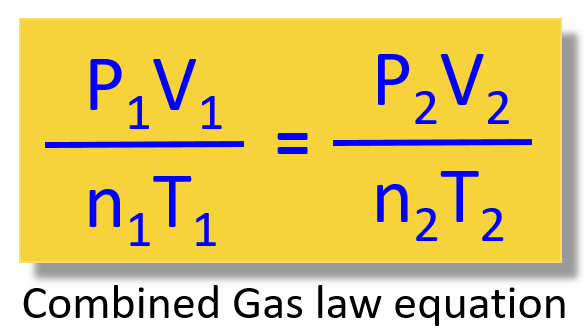
How to Know Which Gas Law Equation to Use - Chemistry Steps
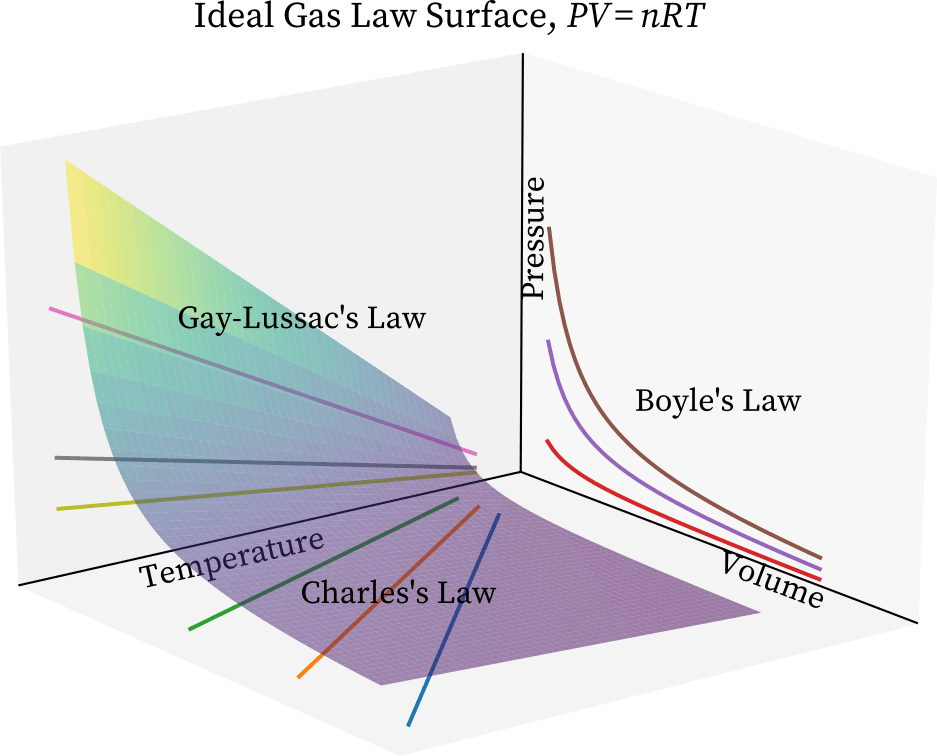
Universal Gas Law Study Guide - Inspirit Learning Inc
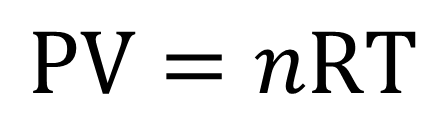
Ideal Gas Rulebreakers - HVAC School

Combined Gas Law CK-12 Foundation
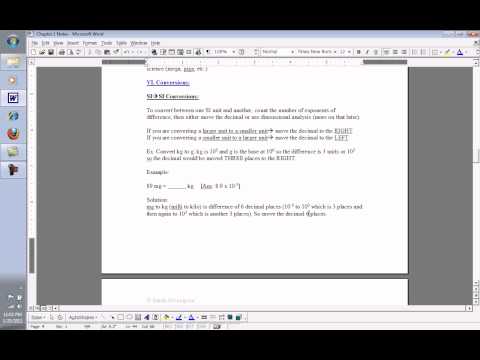
SI Units
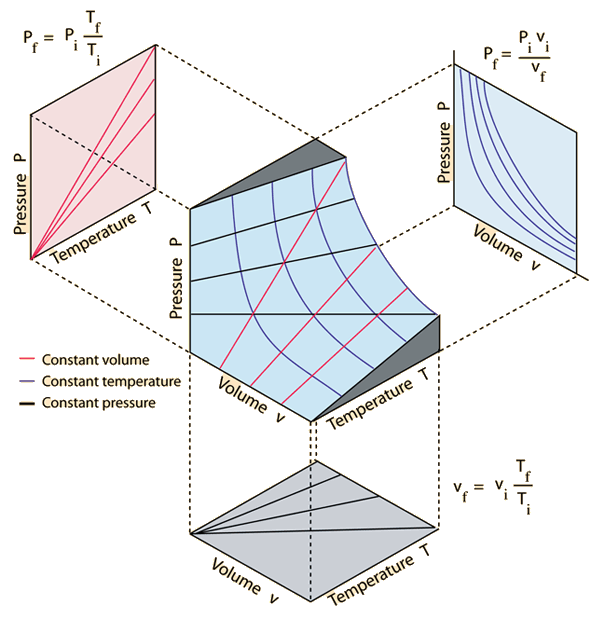
Ideal Gas Law

Hess' Law
Related searches
©2016-2024, doctommy.com, Inc. or its affiliates