Saturday, Oct 05 2024
Ideal Gas Assumptions - Kinetic Theory

By A Mystery Man Writer
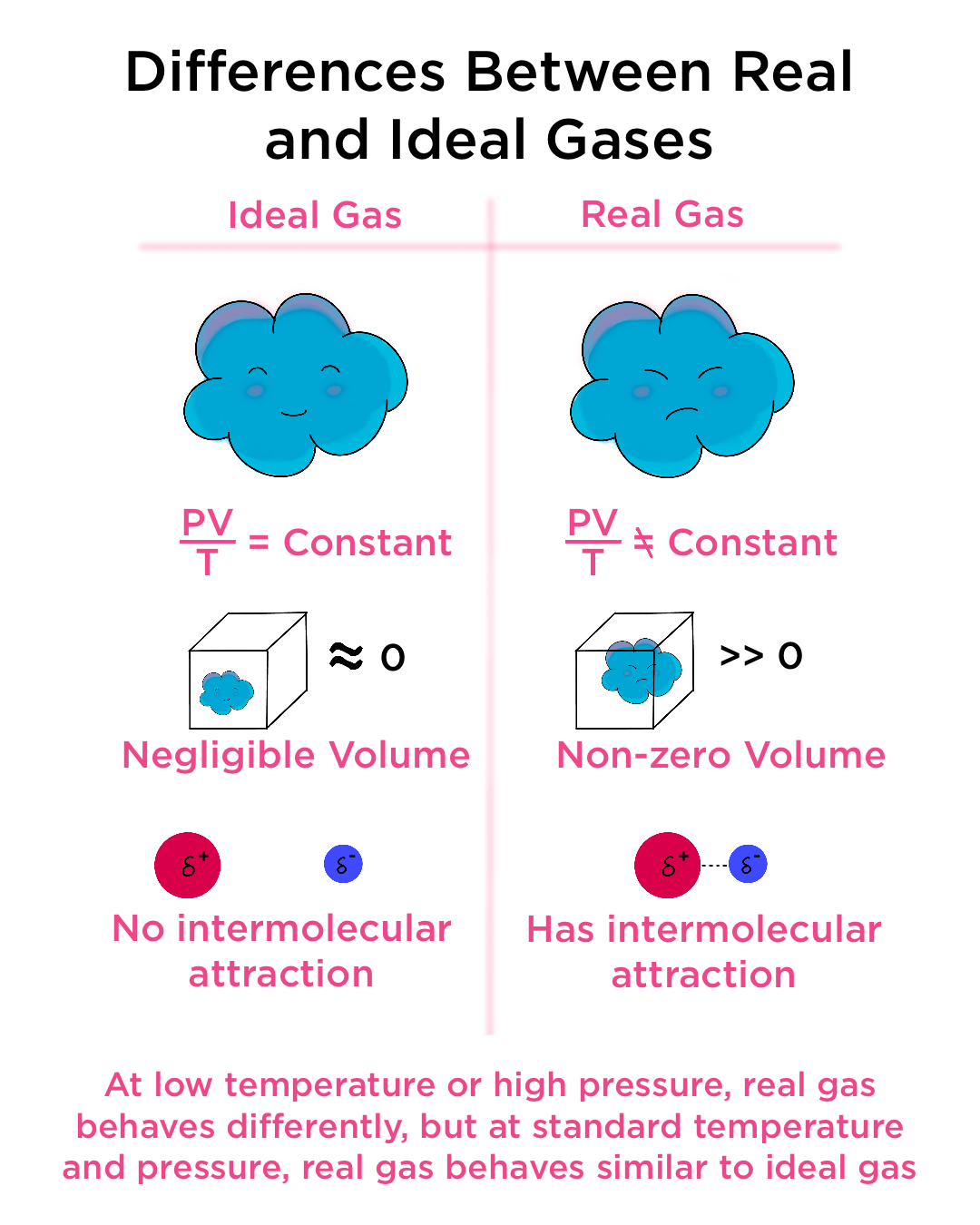
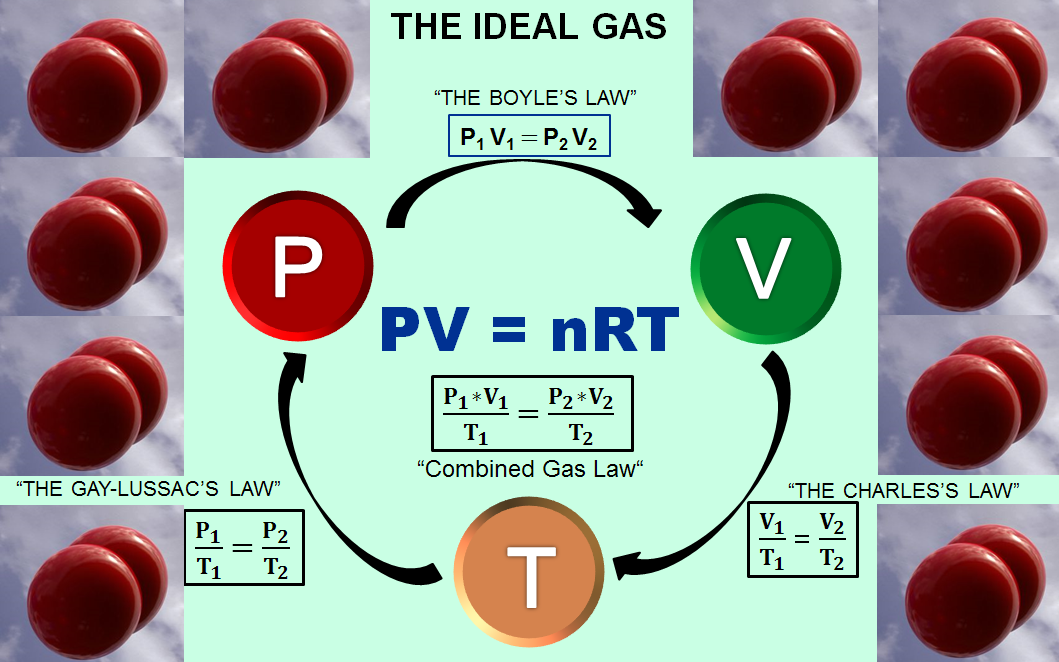
When considering a gas as an ideal gas and applying the ideal gas law pV=nRT, we need to make 4 assumptions. (1) The volume of a molecule within the gas is n

Kinetic Molecular Theory of Gases

1 Ch 10.1 Kinetic Theory: 5 assumptions 1.small particles - far
CBSE Class 11 Physics Kinetic Theory Of Gases Notes

13.8 The Kinetic Molecular Theory of Gases - ChemistrySAANguyen

Assumptions of kinetic theory of gases, pressure exerted by ideal gases, rms velocity for class XI

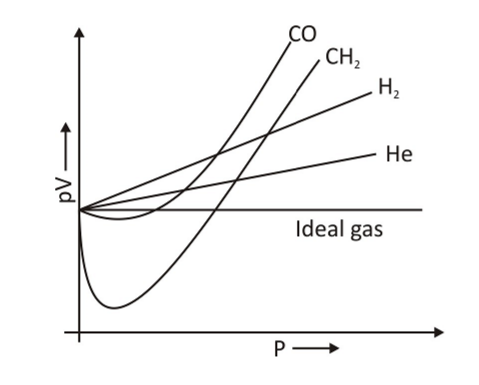
Mechanical model of gas pressure

vt.physics

Kinetic Theory of an Ideal Gas: Equation, Assumption, Concept
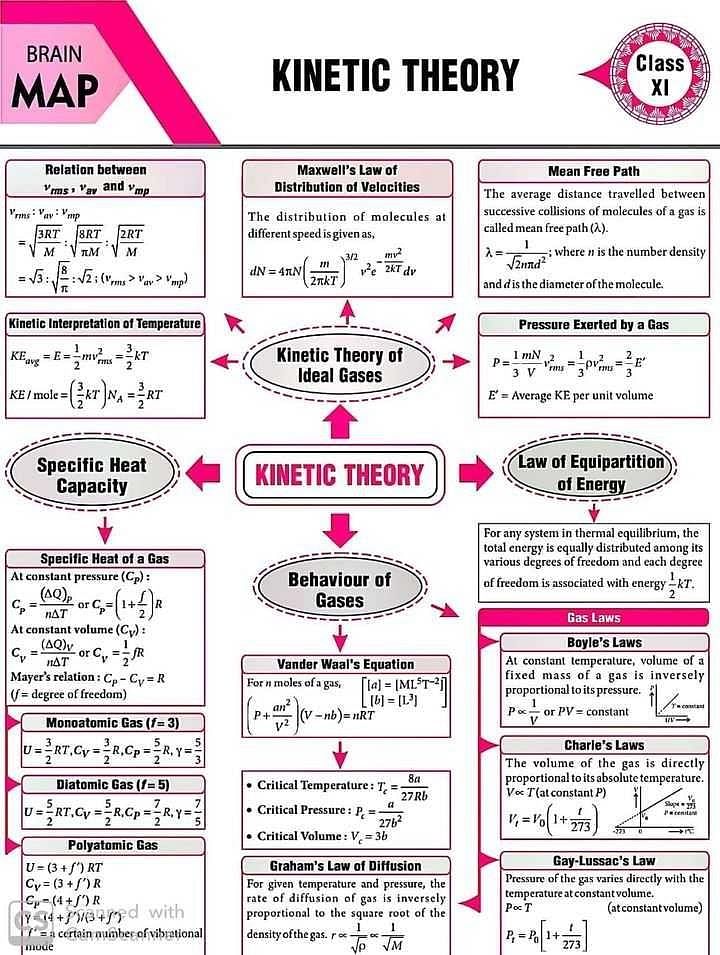
Concept map for kinetic theory ? - EduRev Class 11 Question
Related searches
©2016-2024, doctommy.com, Inc. or its affiliates