10.9: Real Gases - Deviations from Ideal Behavior - Chemistry LibreTexts
By A Mystery Man Writer
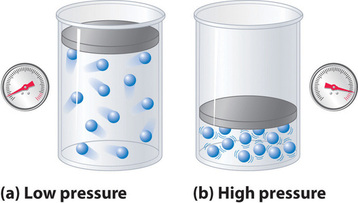
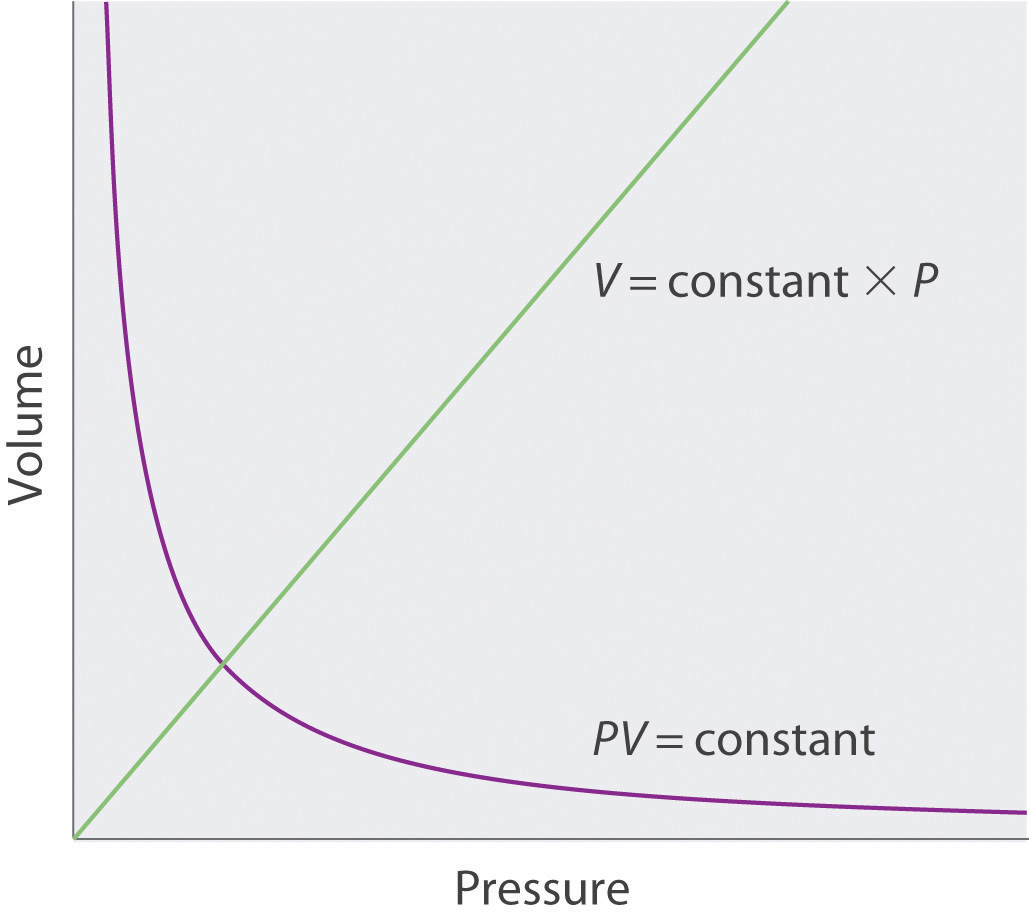
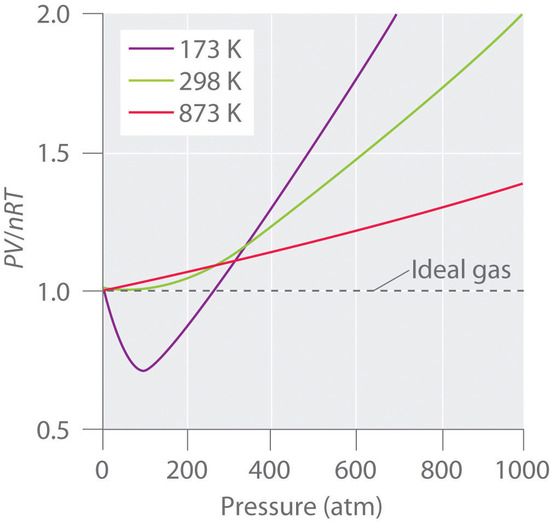
No real gas exhibits ideal gas behavior, although many real gases approximate it over a range of conditions. Gases most closely approximate ideal gas behavior at high temperatures and low pressures. …
No real gas exhibits ideal gas behavior, although many real gases approximate it over a range of conditions. Gases most closely approximate ideal gas behavior at high temperatures and low pressures. Deviations from ideal gas law behavior can be described by the van der Waals equation, which includes empirical constants to correct for the actual volume of the gaseous molecules and quantify the reduction in pressure due to intermolecular attractive forces.
10.E: Exercises - Chemistry LibreTexts

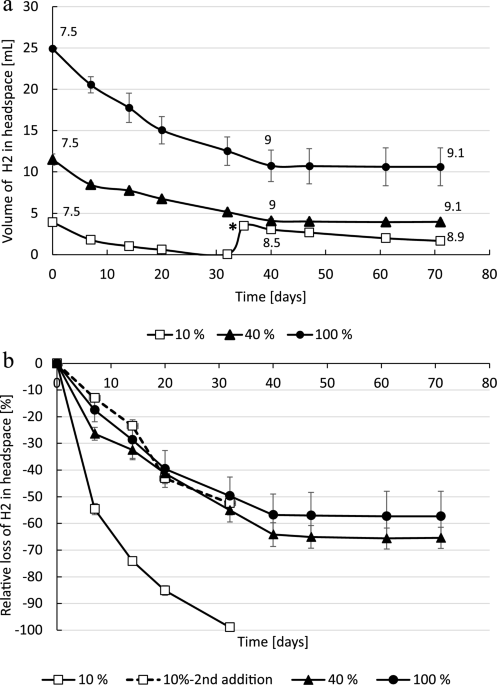
Deviations from Ideal Gas Law Behavior

Chapter 10 Gases. - ppt video online download
10.8: Molecular Effusion and Diffusion - Chemistry LibreTexts
10.9: Real Gases - Deviations from Ideal Behavior - Chemistry LibreTexts

4.2: Real Gases (Deviations From Ideal Behavior) - Chemistry LibreTexts

Lab Report 12 - Ryenna Daly Lab Partner: Kiana TA: Orion Banks 5-5-18 Physical Behavior of Gases Introduction: The physical behavior of gases has four

1.3 Deviation from ideal gas behaviour

Chapter 10 Gases. - ppt download
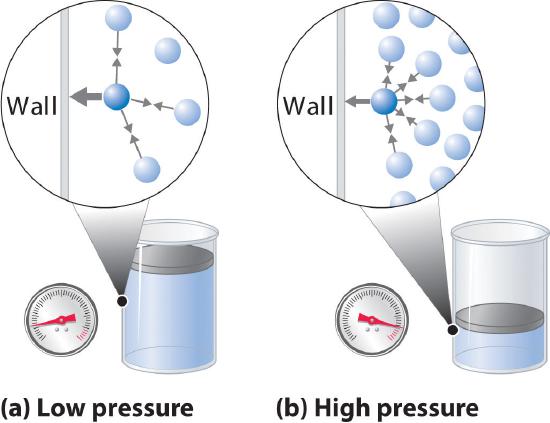
10.8: Real Gases - Chemistry LibreTexts
- ICIW Toppe På Udsalg - Everyday Seamless LS Wmn Dame Hvide

- Warners® Blissful Benefits Dig-Free Comfort Waist with Lace Cotton

- Sanrio Hello Kitty Anime Cartoon Babes Women Stocking Fishnet Hollow Breathable Black Silk Pantyhose Sexy Stockings Girls Gift

- Electric unicycle euc power pads jump pads clarkpads

- Skin Hugging Slim-Fit Yoga Leggings with Inner Pocket