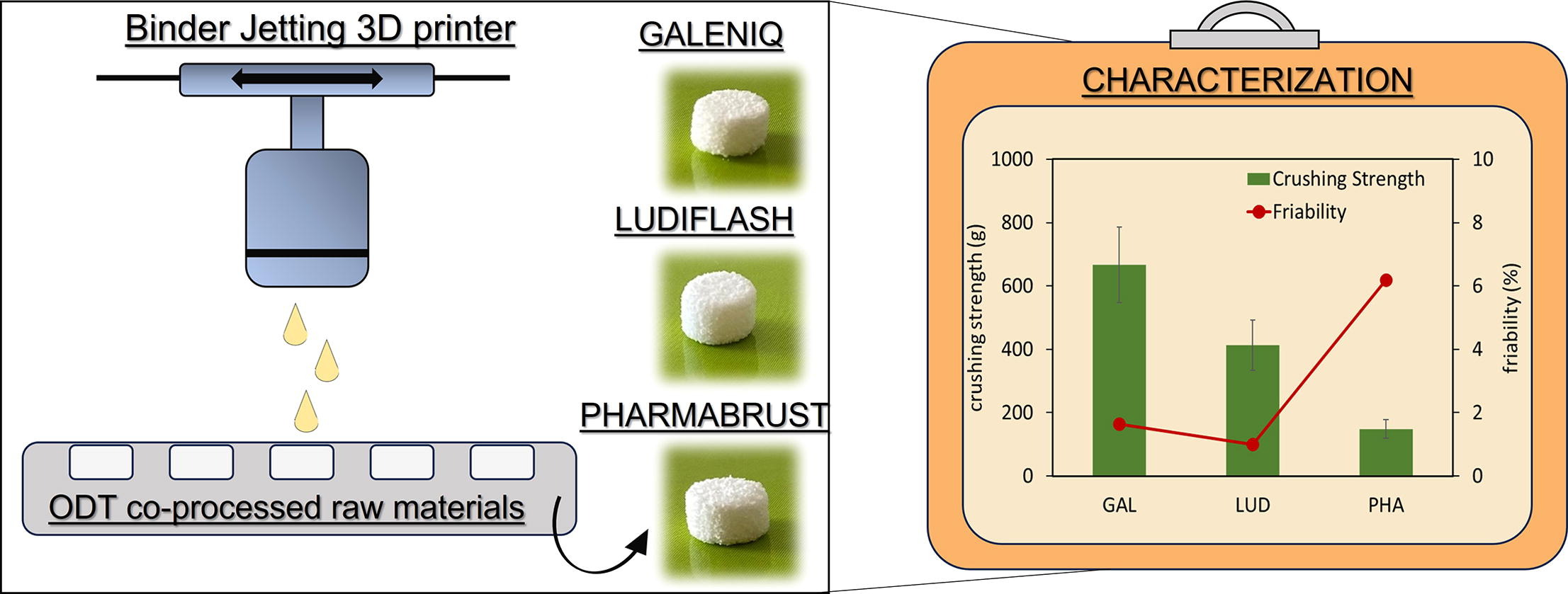
Co-processed materials testing as excipients to produce Orally
By A Mystery Man Writer
The study aimed to estimate the use of commercially available co-processed excipients, conventionally applied in compression protocols.
The use of co-processed materials for Orally Disintegrating Tablets (ODT) preparation by direct compression is well consolidated. However, the evaluation of their potential for ODT preparation by 3D printing technology remains almost unexplored. The

Co-processed materials testing as excipients to produce Orally Disintegrating Tablets (ODT) using binder jet 3D-printing technology - ScienceDirect

Coated dextrin microcapsules of amlodipine incorporable into orally disintegrating tablets for geriatric patients.

Saliha MOUTAHARRIK, PostDoc Position, Bachelor of Industrial Pharmacy, University of Milan, Milan, UNIMI, Department of Pharmaceutical Sciences (DISFARM)

Lucia SALVIONI, PhD, Università degli Studi di Milano-Bicocca, Milan, UNIMIB, Department of Biotechnology and Biosciences

PDF) Development and evaluation of novel, multifunctional co-processed excipients for direct compression of paracetamol tablets

Coated dextrin microcapsules of amlodipine incorporable into orally disintegrating tablets for geriatric patients.

Coated dextrin microcapsules of amlodipine incorporable into orally disintegrating tablets for geriatric patients - ScienceDirect

Luca PALUGAN, Researcher, University of Milan, Milan, UNIMI, Department of Pharmacological Sciences

Co-processed materials testing as excipients to produce Orally Disintegrating Tablets (ODT) using binder jet 3D-printing technology - ScienceDirect

Co-processed Excipients for Pharmaceutical Applications…

Marco GIUSTRA, PhD PostDoc Position, Chemical Sciences and Technologies, Università degli Studi di Milano-Bicocca, Milan, UNIMIB, Department of Biotechnology and Biosciences

Pharmaceutics, Free Full-Text

Co processed excipient

Benefits of Co-processed High-Functionality Excipients in a Continuous Direct Compression Process - 2nd APV Continuous Manufacturing Conference - Pharma Excipients