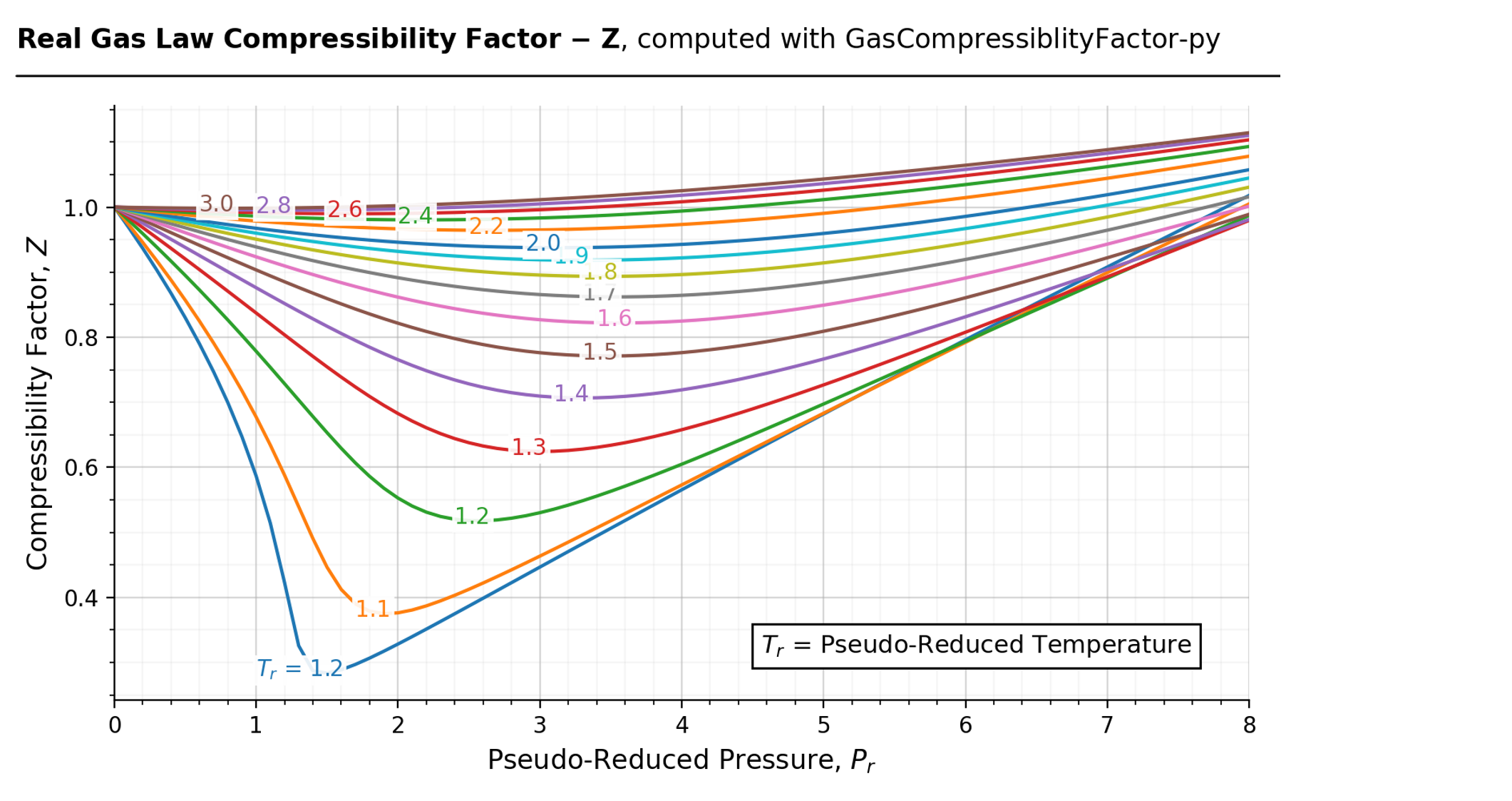
In the following compressibility factor (Z) vs. pressure graph 300 K, the compressibility of CH_{4} pressure < 200 bar deviates from ideal behaviour becauseThe molar volume of CH_{4} is than its molar

By A Mystery Man Writer
Click here:point_up_2:to get an answer to your question :writing_hand:in the following compressibility factor z vs pressure graph at 300 k the compressibility of
Click here👆to get an answer to your question ✍️ In the following compressibility factor -Z- vs- pressure graph 300 K- the compressibility of CH-4- pressure - 200 bar deviates from ideal behaviour becauseThe molar volume of CH-4- is than its molar volume in the ideal stateThe molar volume of CH-4- is than its molar volume in the ideal stateThe molar volume of CH-4- is same as that in its ideal stateIntermolecular interactions between CH-4- molecules decreases

The following graph is plotted between compressibility factor Z versus pressure of a gas at different temperatures.Which of the following statements is /are correct?

Compressibility Factor Z Important Concepts and Tips for JEE Main

New explicit correlation for the compressibility factor of natural gas: linearized z-factor isotherms
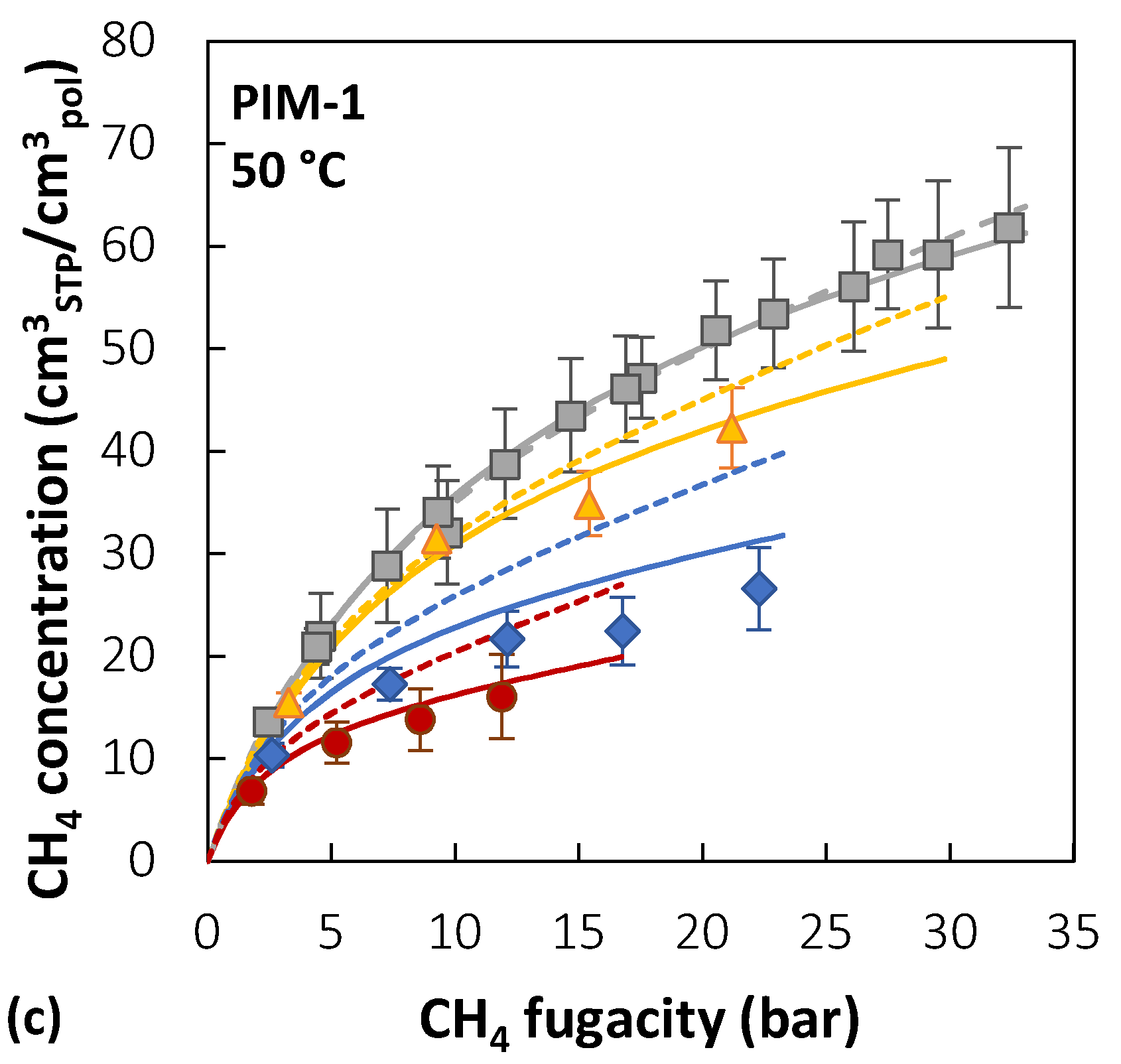
Membranes, Free Full-Text

ars.els-cdn.com/content/image/3-s2.0-B978012803188
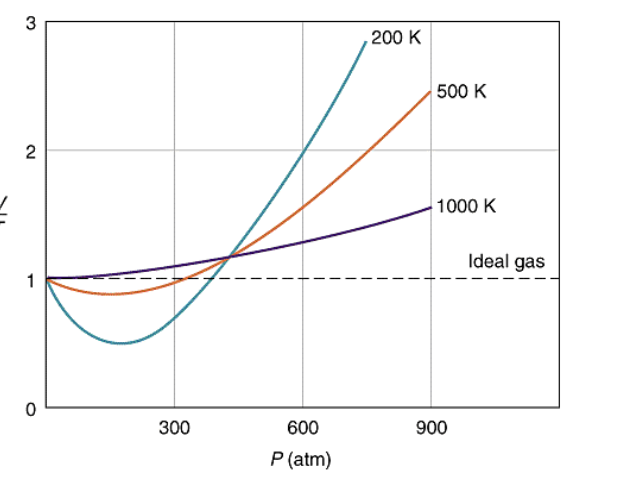
Solved 1. The plot below shows how compressibility factor

Adoption of machine learning in estimating compressibility factor for natural gas mixtures under high temperature and pressure applications - ScienceDirect
States of Matter Class 11 Notes CBSE Chemistry Chapter 5 [PDF]

Non-Ideal Gas Behavior Chemistry: Atoms First