Ranking The Bulkiness Of Substituents On Cyclohexanes: A-Values
By A Mystery Man Writer
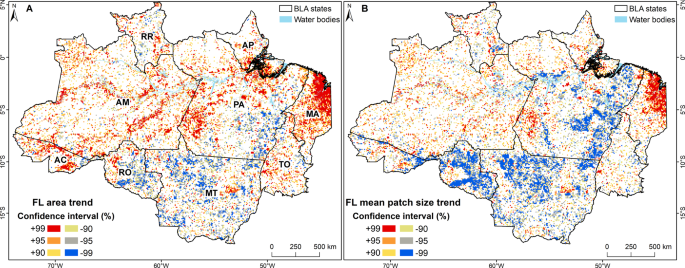
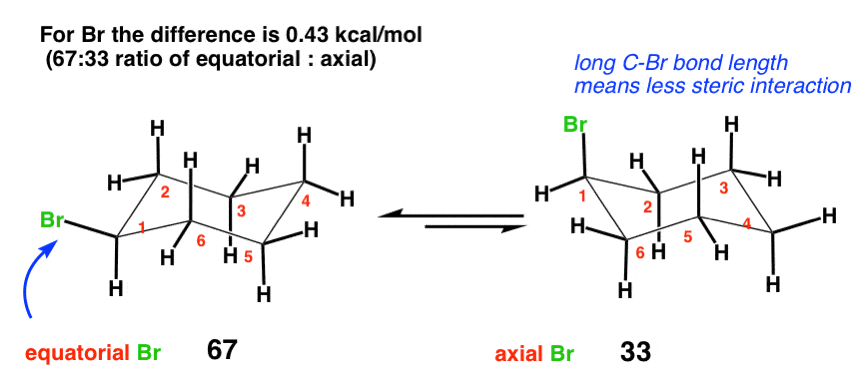
The "steric influence" (or "bulk") of groups on a cyclohexane is quantified by measuring the equilibrium btw axial and equatorial. This is the "A-value"
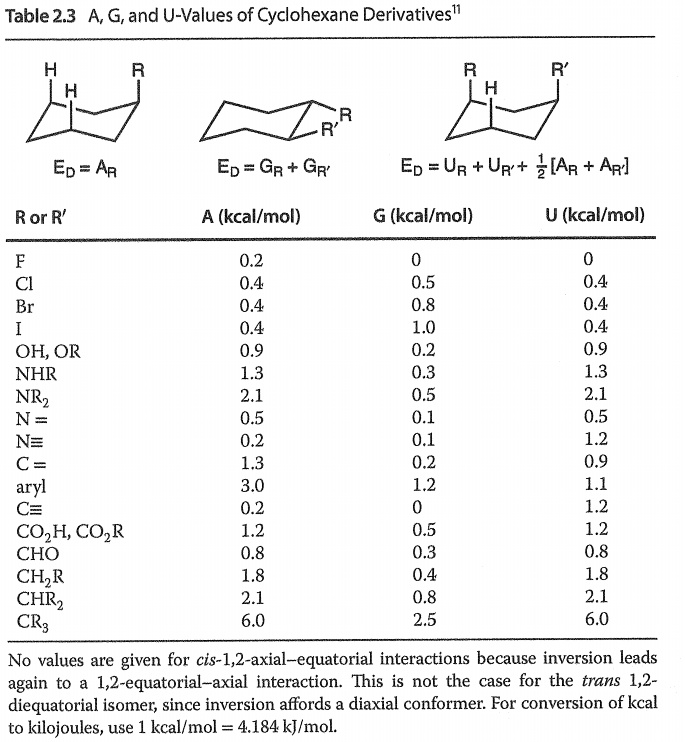
SOLVED: Table 2.3: AG and U-Values of Cyclohexane Derivatives Ep = Ar Ed = Gr + Gr' Ep = Ur Up' + % [Ar + Aq] R or R' A (kcal/mol) (kcal/mol)
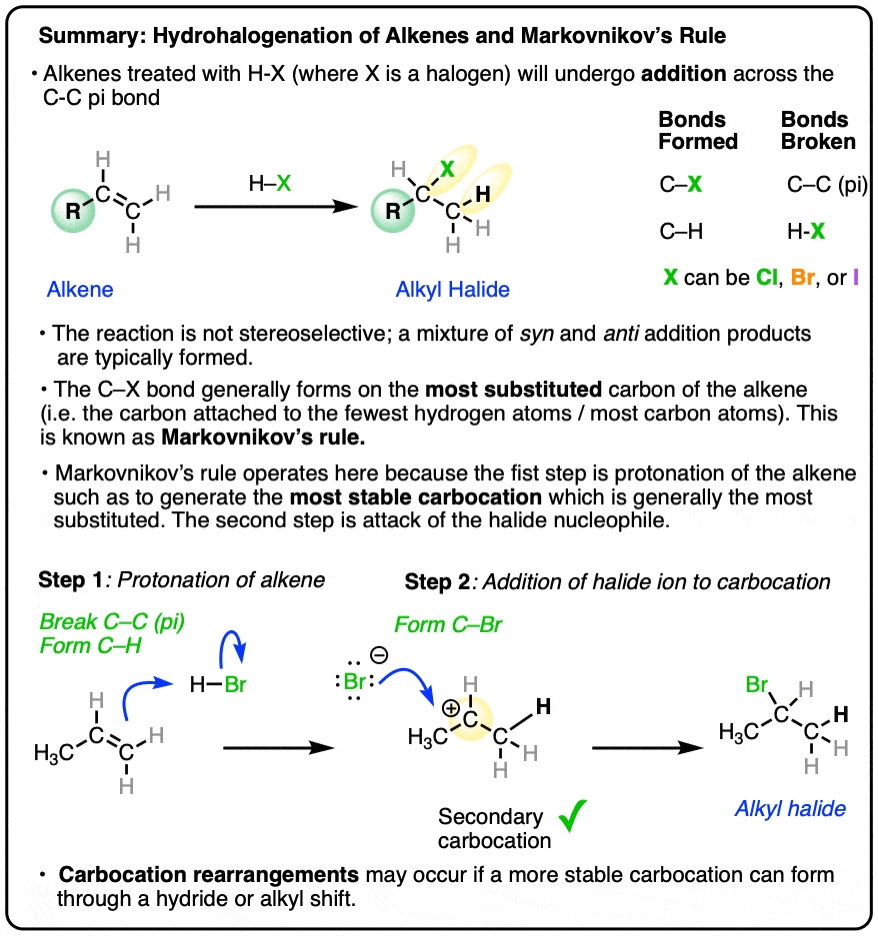
Hydrohalogenation of Alkenes and Markovnikov's Rule – Master
a) ¹H-NMR of tBu-1· in benzene-d6 (top) in equilibrium with tBu-12 in

PDF) Experimental determination of the conformational free energies (A values) of fluorinated substituents in cyclohexane by dynamic 19F NMR spectroscopy. Part 1. Description of the method for the trifluoromethyl group

PDF) Evaluation of quantum chemistry calculation methods for conformational analysis of organic molecules using A-value estimation as a benchmark test
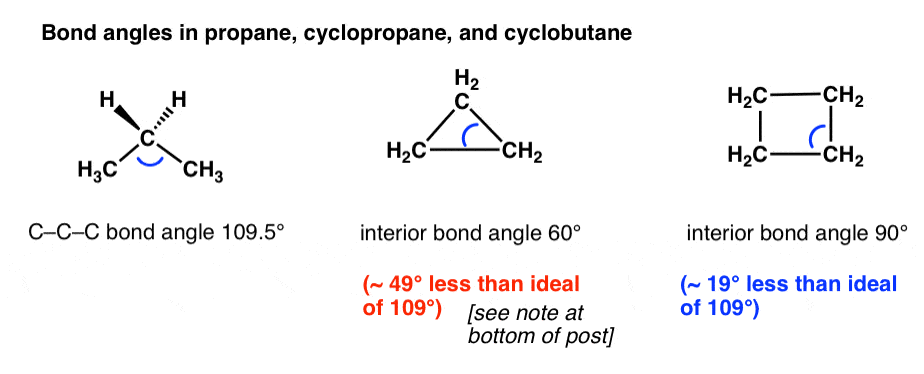
Cycloalkanes - Ring Strain In Cyclopropane And Cyclobutane
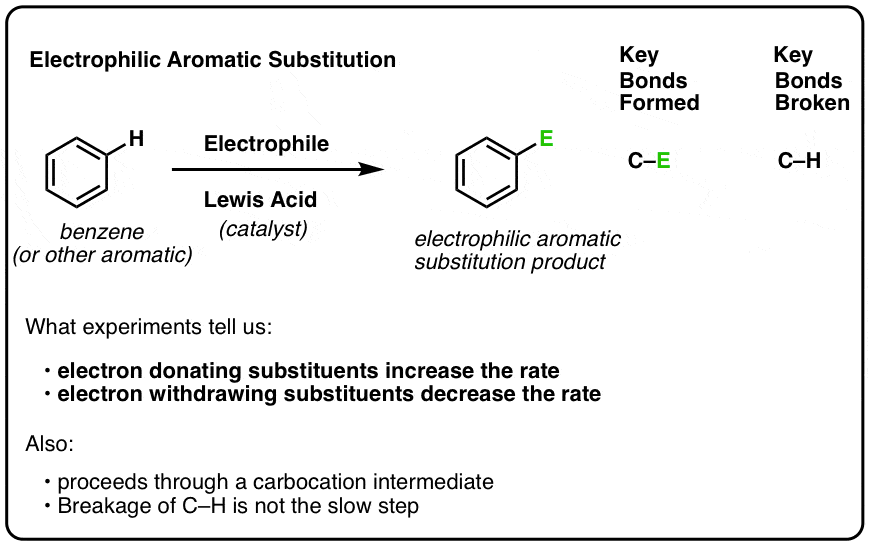
Electrophilic Aromatic Substitution Mechanism – Master Organic

Calculating Energy Difference Between Chair Conformations - Video Tutorials & Practice Problems
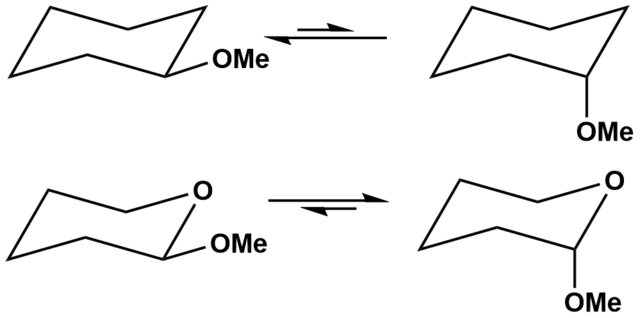
Anomeric effect - Wikipedia
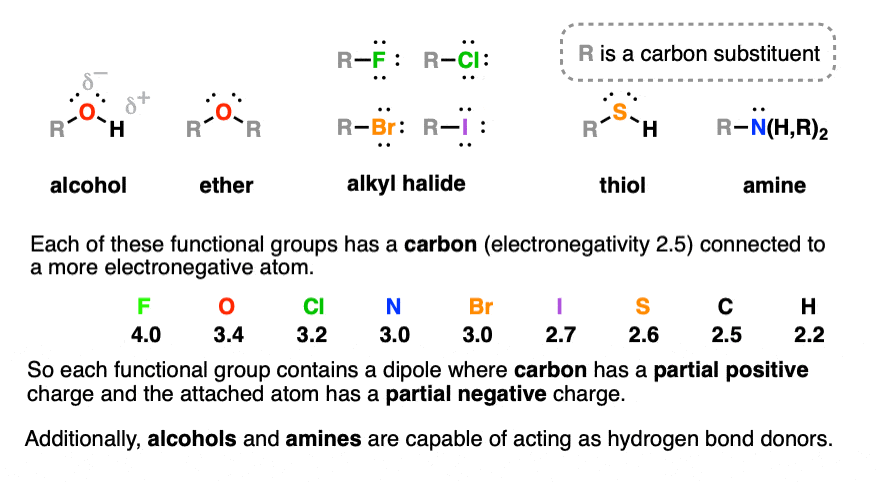
Functional Groups In Organic Chemistry
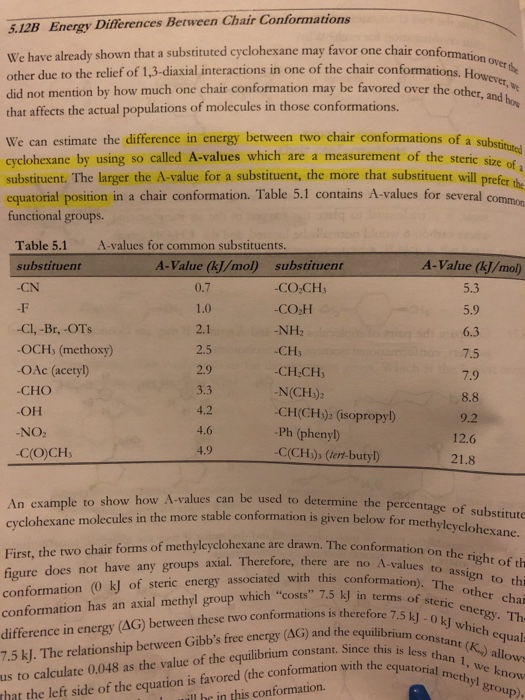
Solved contormation. 5.34 Draw the two chair conformations

Solved A-values represent the amount of energy required to