SOLVED: Problem 1: Calculate the compressibility factor and the molar volume for ethane at 125°C and 24 bar using: i. The generalized Pitzer correlation using Lee and Kesler tables. ii. The Pitzer

By A Mystery Man Writer
VIDEO ANSWER: There is a chance that the pressure will be 300 bar and the temperature will be zero degree centigrade, so it will be a temperature of 3700 degrees. The T is equal to the number of moles. R is the universal gas constant, and the ideal
Numerade is a venture-backed, high-growth education technology startup based in Pasadena. We are singularly focused on creating exceptional video and interactive content experiences for education making the knowledge and skills of world class educators widely accessible and affordable to student audiences of all backgrounds. Our mission is to close the educational opportunity gap by unlocking and democratizing access to extraordinary educators and the content they have to offer.
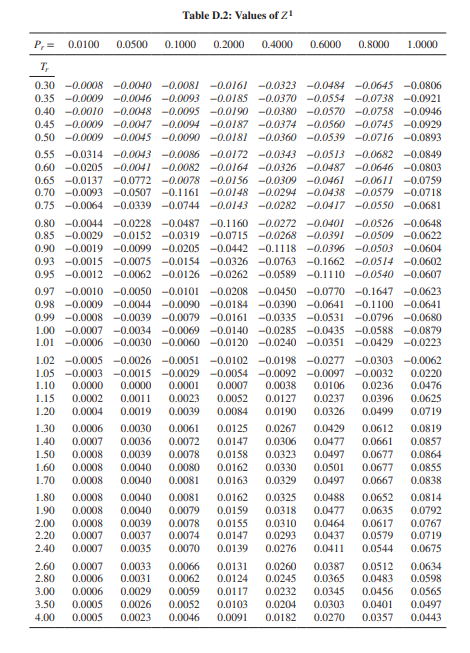
Solved Use the Pitzer correlation in Z (using Lee/Kester

Solved Use the Pitzer correlation in Z (using Lee/Kester

Study of Liquid−Liquid and Liquid−Liquid−Vapor Equilibria for Crude Oil Mixtures with Carbon Dioxide and Methane Using Short- Wave Infrared Imaging: Experimental and Thermodynamic Modeling

Calculate Z and V for ethylene at 25°C and 12 bar by the following equations: (a) The truncated virial

Study of Liquid−Liquid and Liquid−Liquid−Vapor Equilibria for Crude Oil Mixtures with Carbon Dioxide and Methane Using Short- Wave Infrared Imaging: Experimental and Thermodynamic Modeling

Solved Calculate the compressibility factor Z and the molar

Binary interaction parameters for nonpolar systems with cubic equations of state: a theoretical approach 1. CO2/hydrocarbons using SRK equation of state

Solved The Pitzer method is a common methodology used to

Calculate Z and V for ethylene at 25°C and 12 bar by the following equations: (a) The truncated virial

SOLVED: Using the Pitzer correlations by both Lee/Kesler and Abbott, as well as the SRK EOS, calculate Z and V for the following substances: a) Butane at 500K and 9 bar. b)

Binary interaction parameters for nonpolar systems with cubic equations of state: a theoretical approach 1. CO2/hydrocarbons using SRK equation of state

Solved Use the compressibility charts to answer the

SOLVED: Problem 1: Calculate the compressibility factor and the molar volume for ethane at 125°C and 24 bar using: i. The generalized Pitzer correlation using Lee and Kesler tables. ii. The Pitzer

Chapter 2: Lee/Kesler Example 1
- Compressibility Factor Calculator

- Compressibility Calculator - Calculator Academy
- At low pressure, the van der waal's equation is written as (P+ a/V^2)V=RT . Then compressibility factor is then equal to
- Compressor and jet vacuum system:, by Maryambotshekan

- Which of the following statements is/are correct? (a) all real gases are less compressible

- XS Wild Fable patchwork bomber coat Blue Denim V St oversized jean Jacket

- Women's Cottonique W12227 Plus Latex Free Organic Cotton Front

- US Sexy Women's Lingerie Set Lace Swimwear Bikini Bra G-String Thongs Beachwear

- Body Shaper Under Pants Shapewear Under Dresses - Slimming Body Shaper for Postpartum Mother Belly and Hulzogul Hip Shaper : : Clothing, Shoes & Accessories

- 320+ Grated Orange Zest Stock Photos, Pictures & Royalty-Free Images - iStock
